
Environmentally Friendly and Facile Synthesis of 2-oxo- and Thioxo-1,2,3,4-tetrahydropyrimidines Catalyzed by Formic Acid as a Natural Green and Bio-based Catalyst under Solvent-Free Conditions
Farzaneh Mohamadpour*Published Date : 2019-12-09
DOI : ttps://doi.org/10.12982/CMUJNS.2020.0005
Journal Issues : Number 1, January-March 2020
ABSTRACT A green synthetic route to the facile Biginelli synthesis of 2-oxo- and thioxo-1,2,3,4-tetrahydropyrimidines have been developed using formic acid as a natural green and bio-based catalyst under solvent-free reaction conditions. All reactions are completed in a short period of times and the products are obtained in high to excellent yields. The salient features of this green approach are simple work-up with no necessity of chromatographic purification steps, absence of hazardous organic solvents, use of safe, non-volatile, noncorrosive and readily green catalyst, solvent-free conditions, one-pot reaction, eco-friendly and clean synthesis.
Keywords: Formic acid, Natural green and Bio-based catalyst, 2-oxo- and Thioxo-1,2,3,4-tetrahydropyrimidines, Solvent-free conditions
INTRODUCTION
Synthesis of heterocyclic compounds has attracted great interests due to their wide applicability in life and nature. The compounds with pyrimidinone derivatives are reported as, such as calcium channel blockers, α-1a-antagonists (Prakash et al., 2008), mitotic kinesin Eg5 inhibition (Kapoor et al., 2000), anti cancer (Mal3-101) (Wisen et al., 2008), anti HIV agent (Heys et al., 2000), antibacterial and antifungal (Ashok et al., 2007), antiviral (Hurst et al., 1961), antioxidative (Magerramow et al., 2006). The representatives such as batzelladines, ptilomycalines and crambescidines exhibit many biological activities such as anticancer, antifungal, anti HIV, etc (Bewley et al., 2004).
One of the dominating factors in recent organic synthetic routs is green chemistry. Atom economy, reduction in byproduct, number of steps in organic synthesis, energy cost, produced waste, use of non-hazardous reagents in catalytic protocols are one of the most important goals of green chemistry. Furthermore, organic reactions under solvent-free conditions for green and clean synthesis of organic compounds have attracted much interest in organic chemists. Due to, our recent studies focused on developing of green catalyst in multi-component reactions (Mohamadpour et al., 2016; Mohamadpour et al., 2017; Lashkari et al., 2018; Mohamadpour., 2018a; Mohamadpour et al., 2018b; Mohamadpour et al., 2018c; Mohamadpour., 2019).
In recent decades, a number of methodologies for the preparation of these compounds have been reported that is including various catalysts such as calcium fluoride (Chitra et al., 2009), copper(II)sulfamate (Liu et al., 2008), baker’s yeast (Kumar et al., 2007), hydrotalcite (Lal et al., 2012), hexaaquaaluminium (III) tetrafluoroborate (Litvic et al., 2010), TBAB (Ahmad et al., 2009), copper (II) tetrafluoroborate (Kamal et al., 2007), Copper (II) acetate (Khodja et al., 2014), [Btto][p-TSA] (Zhang et al., 2015), triethylammonium acetate (Attri et al., 2017), p-dodecylbenzenesulfonic acid (Aswin et al., 2014), TMSPTPOSA (Rao Jetti et al., 2017), hierarchical zeolite (Shahid et al., 2017), bismuth(III)nitrate or PPh3 (Slimi et al., 2016), lanthanum oxide (Kuraitheerthakumaran et al., 2016) and dendrimer-PWA (Safaei-Ghomi et al., 2018). Some of these methodologies have limitations such as difficult work-up, toxic and expensive catalysts, low yields, the use of strongly acidic conditions and long time reactions. As part of our ongoing research program on the development of green methodologies, herein, we report a green and facile one-pot synthesis of synthesis of 2-oxo- and thioxo-1,2,3,4-tetrahydropyrimidines via three-component Biginelli (Biginelli et al., 1893) reaction between β-keto esters, aldehyde derivatives and urea/thiourea in the presence of catalytic amount of formic acid under thermal and solvent-free conditions. The advantages of formic acid as a mild, natural green and bio-based acidic catalyst (Thompson et al., 1997) in organic synthesis are environmentally friendly, highly efficient and non-toxic. It is noted that, formic acid as a catalyst shows superior properties like commercially available. It is inexpensive and can be easily handled. The merits of this acidic catalyst does not end here, in this present work, the products were obtained through simple filtering with no need column chromatographic separation. Furthermore, high to excellent yields, short reaction times and eco-friendly procedure that makes our protocol alternative in comparison to some of the earlier reported methods.
MATERIALS AND METHODS
General
Melting points all compounds were determined using an Electro thermal 9100 apparatus. 1H NMR spectra were recorded on a Bruker DRX-400 Avance instrument with DMSO-d6 as solvents. All reagents and solvents were purchased from Merck, Fluka and Acros chemical companies were used without further purification.
General procedure for preparation of 2-oxo- and thioxo-1,2,3,4 tetrahydropy-rimidines (4a- q). A mixture of aldehyde derivatives (1, 1.0 mmol) and urea/thiourea (2, 1.5 mmol), ethyl/methyl acetoacetate (3, 1.0 mmol) was heated under solvent-free conditions at 70 °C for appropriate time in the presence of formic acid (10 mol %). After completion of the reaction (by thin layer chromatography TLC) the mixture was cooled to rt and cold water was added and the precipitated was separated by filtration and recrystallized from ethanol to afford the pure products (4a- q).
Spectra data some of known products are represented below:
5-Ethoxycarbonyl-6-methyl-4-(4-nitrophenyl)-3,4-dihydropyrimidin-2(1H)-one (4a):
Yield: 87%; m.p. 206-208 °C; 1H NMR (400 MHz, DMSO-d6): 1.10 (3H, t, J= 9.6 Hz, CH3CH2), 2.28(3H, s, CH3), 3.99 (2H, q, J=9.2 Hz, CH2O), 5.27 (1H, s, Hbenzylic), 7.50-7.53 (2H, m, HAr), 7.23 (2H, d, J= 9.2Hz, HAr), 7.92 and 9.38 (2H, 2s, 2NH).
5-Methoxycarbonyl-6-methyl-4-(2-chlorophenyl)-3,4-dihydropyrimidin-2(1H)-one (4c):
Yield: 80%; m.p. 250-252 °C; 1H NMR (400 MHz, DMSO-d6): 2.31 (3H, s, CH3), 3.46 (3H, s, OCH3), 5.62 (1H, s, Hbenzylic), 7.28-7.34 (3H, m, HAr), 7.42 (1H, d, J=7.2 Hz, HAr), 7.72 and 9.36(2H, 2s, 2NH).
5-Ethoxycarbonyl -6-methyl -4-(4-methoxyphenyl)-3,4-dihydropyrimidin-2(1H)–one (4g):
Yield: 86%; m.p. 202-204°C; 1H NMR (400 MHz, DMSO-d6): 1.11 (3H , t, J= 9.6 Hz, CH3CH2), 2.24(3H, s, CH3), 3.73 (3H, s, OCH3), 3.99 (2H, q, J=9.6 Hz, CH2O), 5.09 (1H, s, Hbenzylic), 6.89 (2H, d, J= 8.4Hz, HAr), 7.15(2H, d, J= 8.8Hz, HAr), 7.70 and 9.18 (2H, 2s, 2NH).
5-Ethoxycarbonyl-6-methyl-4-phenyl-3,4-dihydropyrimidin-2(1H)-one (4h):
Yield: 86%; m.p. 198-200 °C; 1H NMR (400 MHz, DMSO-d6): 1.10 (3H, t, J= 7.2 Hz, CH3CH2), 2.26 (3H, s, CH3), 3.99 (2H, q, J=7.2 Hz, CH2O), 5.15 (1H, s, Hbenzylic), 7.26 (3H, d, J= 7.2 Hz, HAr), 7.33 (2H, t, J=7.2 Hz, HAr), 7.76 and 9.21 (2H, 2s, 2NH).
5-Ethoxycarbonyl-6-methyl-4-phenyl-3,4-dihydropyrimidin-2(1H)-thione (4k):
Yield: 85%; m.p. 209-211 °C; 1H NMR (400 MHz, DMSO-d6): 1.11 (3H , t, J= 7.2 Hz, CH3CH2), 2.31 (3H, s, CH3), 4.02 (2H, q, J=7.2 Hz, CH2O), 5.19 (1H, s, Hbenzylic), 7.23 (2H, d, J=7.2 Hz, HAr), 7.28 (1H, t, J=7.2 Hz, HAr), 7.36 (2H, t, J=7.2 Hz, HAr), 9.68 and 10.36 ( 2H, 2s, 2NH).
RESULTS
At the beginning, we performed three-component condensation of benzaldehyde (1.0 mmol), urea (1.5 mmol) and ethyl acetoacetate (1.0 mmol) in the presence of formic acid (10 mol%) under solvent-free at 70 °C, the product 4h was found in 86%, which was confirmed by 1H NMR spectroscopy. Encouraged by this result, we have chosen this reaction as a model reaction to study the reaction conditions further for the synthesis of 2-oxo- and thioxo-1,2,3,4-tetrahydropyrimidines (4a-q). The catalyst plays an important role in the success of the reaction in terms of the rate of the reaction and yields. In order to optimize the reaction conditions, quantity of the catalyst required was determined. No product could be detected in the absence of the catalyst even after 10 h (Table 1, entry 1). Then, 5 mol% formic acid was used to perform the reaction. But it requires slightly longer reaction time and low yields (Table 1, entry 2). Therefore, the loading of catalyst was gradually increased from 5 mol% to 15 mol% (Table 1). It was found that 10 mol% of formic acid are optimal to carry out the reactions in a short duration (Table 1, entry 3). The use of excess of catalyst did not alter either reaction time or yield of the product. Thus, the use of 10 mol% formic acid is ideal to achieve the desired product in high yields. We also investigated different temperatures for the model reaction (Table 1). It was observed that fast reaction occurred on raising the temperature from rt to 80 °C and the yield of preferred production increased significantly (Table 1). We were satisfied to find that the reaction proceeded smoothly and almost complete conversion of reactants was observed at 70 °C to afford the desired product (4h) in 86% yields within 20 min (Table 1, entry 3). A further increase in the temperature did not affect the product yield (Table 1, entry 8). Having optimized reaction conditions, we synthesized a series 2-oxo- and thioxo-1,2,3,4-tetrahydropyrimidines via aryl aldehyde derivatives (1, 1.0 mmol), urea/ thiourea (2, 1.5 mmol) and ethyl/methyl acetoacetate (3, 1.0 mmol) (4a-q) using 10 mol% formic acid as the catalyst under solvent-free conditions at 70 °C (Scheme 1) and the results are summarized in Table 2.
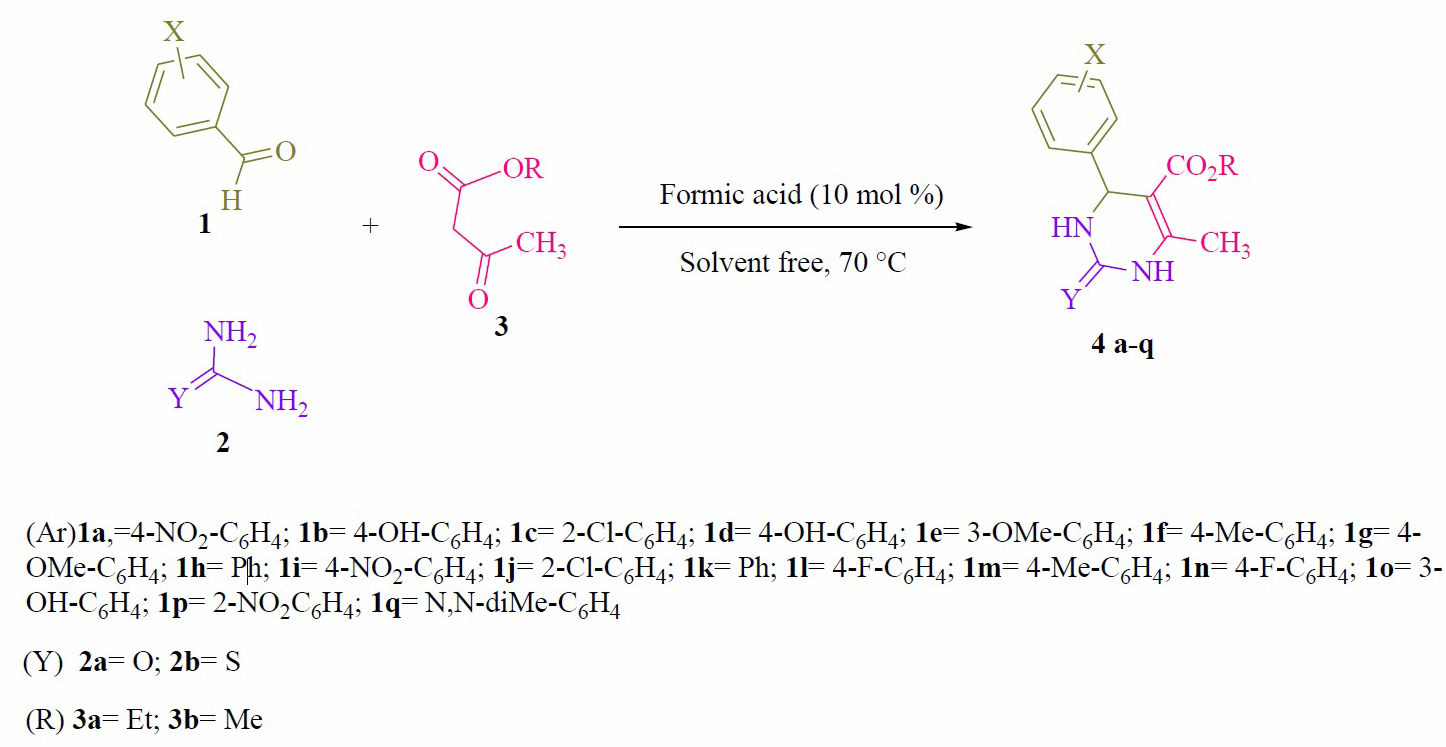
Figure 1. Synthesis of 2-oxo- and thioxo-1,2,3,4-tetrahydropyrimidines.
Table 1. Optimization of the reaction condition on the synthesis of 4h a.
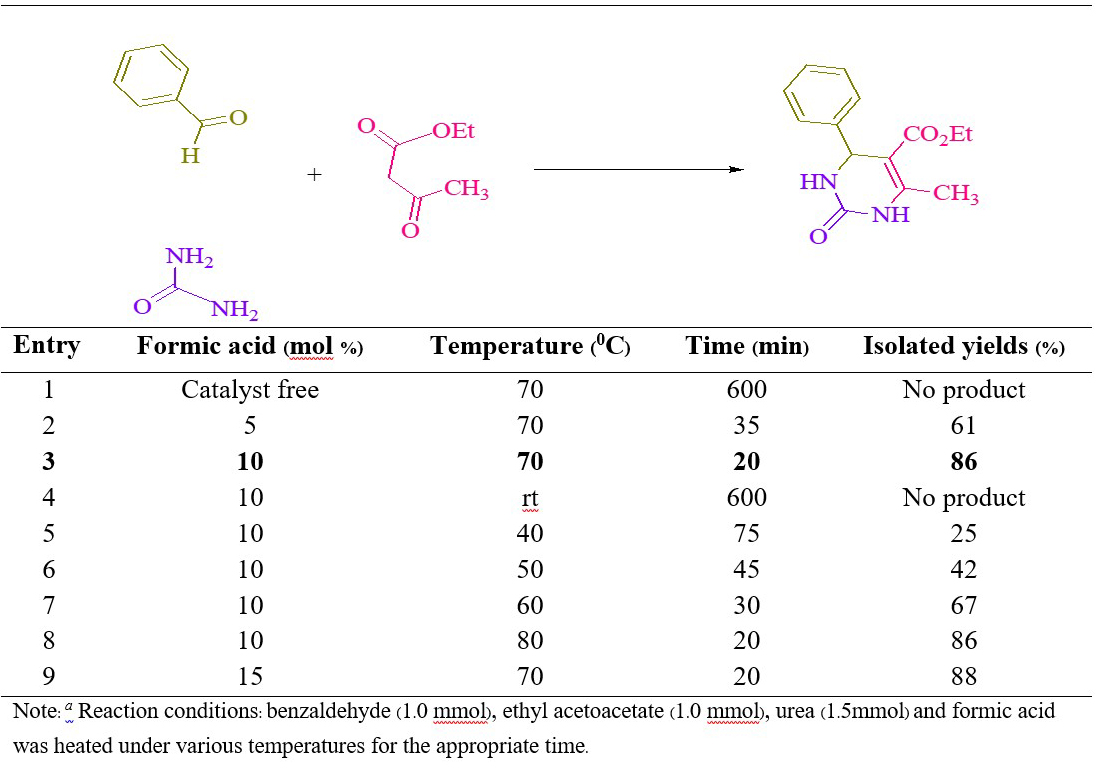
Table 2. Synthesis of 2-oxo- and thioxo-1,2,3,4-tetrahydropyrimidines.
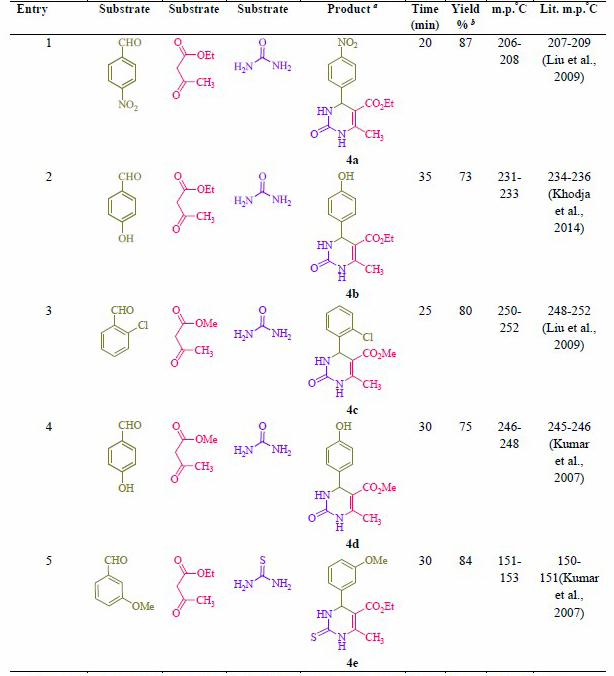
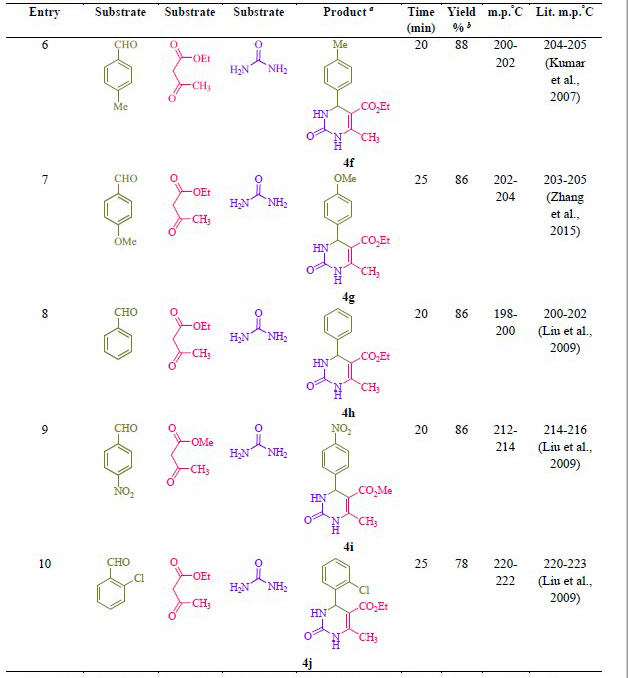
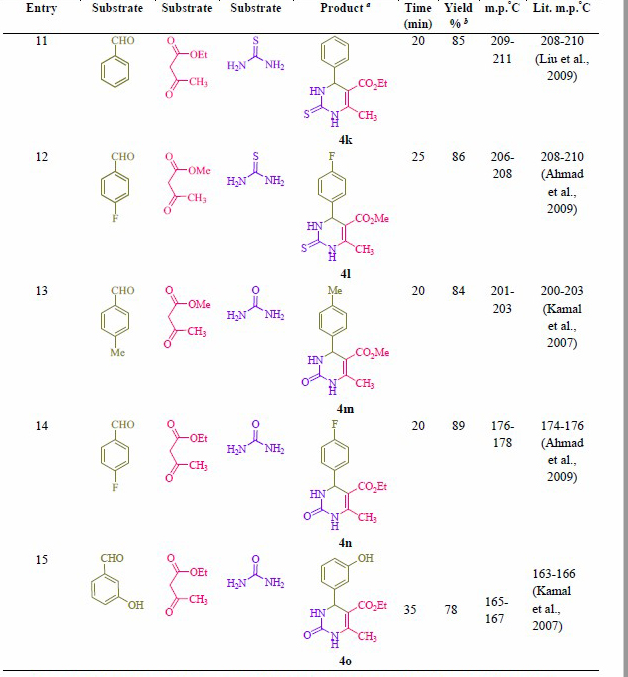
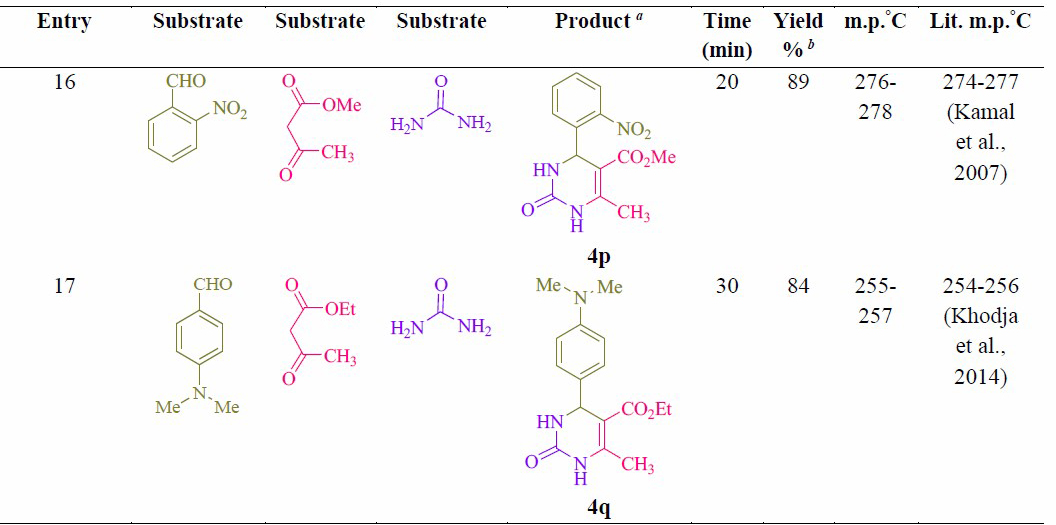
Note: a Isolated yield. b Reaction conditions: Aryl aldehyde derivatives (1.0 mmol), ethyl/methyl acetoacetate (1.0 mmol), urea/thiourea (1.5 mmol) and formic acid (10 mol %) was heated at 70 °C.
DISCUSSION
Although different mechanistic pathways have been previously proposed (Kamal et al., 2007; De Souza et al., 2009; Alvim et al., 2014; Rao Jetti et al., 2017; Safaei-Ghomi et al., 2018), we believe that the reaction may proceed through an initial N-acylimine B formed from aldehyde 3 and urea 2 (Figure 2). The coordination of the lone-pair of the nitrogen atom in the N-acylimine B with the formic acid could lead to the in situ formation of an N-carbamoyliminium ion C, which is sufficiently electrophilic to react with the enol form of ethyl acetoacetate A affording the open chain intermediate D. Further intramolecular cyclization, with elimination of H2O, produce the 2-oxo- and thioxo-1,2,3,4-tetrahydropyrimidines 4.
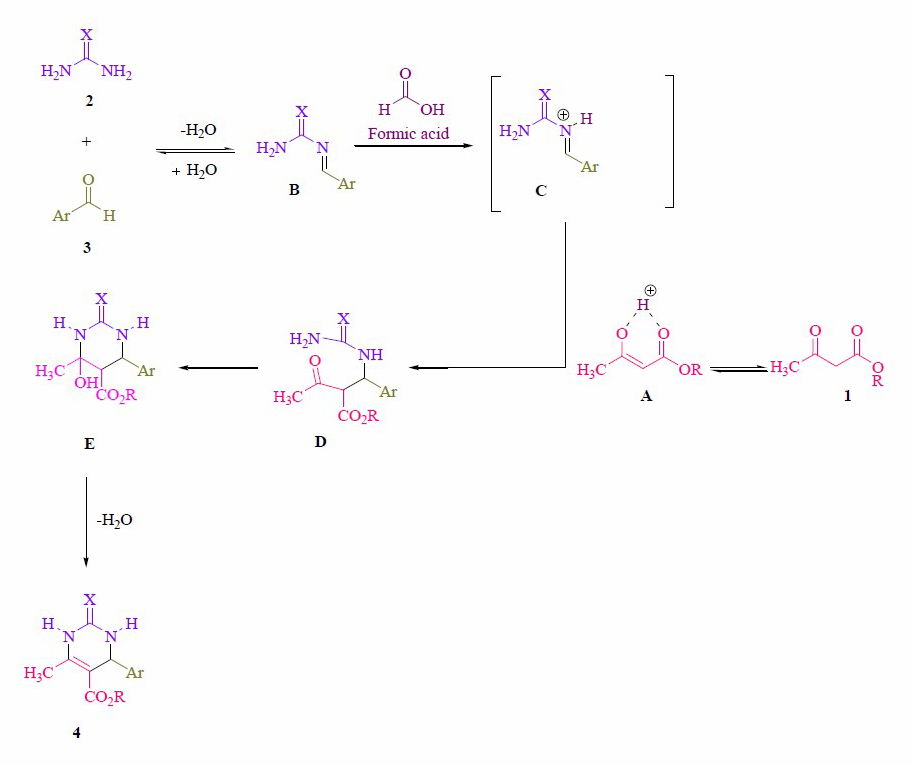
Figure 2. Proposed mechanistic route for the synthesis of 2-oxo- and thioxo-1,2,3,4-tetrahydropyrimidines.
Comparison of catalytic ability some of catalysts reported in the literature for synthesis of 2-oxo- and thioxo-1,2,3,4-tetrahydropyrimidines are shown in Table 3. This study reveals that formic acid has shown its extraordinary potential to be an alternative green, bio-based, readily, highly efficient and inexpensive catalyst for the Biginelli reaction. In Addition, the use of solvent-free conditions with high to excellent yields and short reaction times in the reaction with both urea and thiourea are the notable advantages this green and simple procedure.
Table 3. Comparison of catalytic ability some of catalysts reported in the literature for synthesis of 2-oxo- and thioxo-1,2,3,4-tetrahydropyrimidinesa.
|
References |
Time/Yield (%) |
Conditions |
Catalyst |
Entry |
|
|
(Kumar et al., 2007) |
24h/84 |
Room temperature |
bakers, yeast |
1 |
|
|
(Lal et al., 2012) |
35 min/84 |
Solvent-free, 80 °C |
Hydrotalcite |
2 |
|
|
(Litvic et al., 2010) |
20 h/81 |
MeCN, Reflux |
[Al(H2O)6](BF4)3 |
3 |
|
|
(Kamal et al., 2007) |
30 min/90 |
Room temperature |
Cu(BF4)2.xH2O |
4 |
|
|
(Zhang et al., 2015) |
30 min/96 |
Solvent-free, 90 °C |
[Btto][p-TSA] |
5 |
|
|
(Attri et al., 2017) |
45min/90 |
Solvent-free, 70 °C |
triethylammonium acetate |
6 |
|
|
(Aswin et al., 2014) |
3 h/94 |
Solvent-free, 80 °C |
p-dodecylbenzenesulfonic acid |
7 |
|
|
(Rao Jetti et al., 2017) |
3 h/95 |
EtOH/Reflux |
TMSPTPOSA |
8 |
|
|
This work |
20 min/86 |
Solvent-free, 70 °C |
Formic acid |
9 |
|
|
Note: a Based on the three-component reaction of benzaldehyde, ethyl acetoacetate and urea. |
|||||
CONCLUSION
In summary, a natural green, highly efficient and bio-based acidic catalyst, i.e. formic acid was developed and exploited for clean, facile and economical one-pot synthesis of 2-oxo- and thioxo-1,2,3,4-tetrahydropyrimidines from starting materials under solvent-free conditions. This method gave an insight on the credibility of pathway followed by the aforementioned green and bio-based catalyst in aiding the heterocyclic compound formation. Cleaner reaction profile, simple column-free work up condition, shorter reaction times, high to excellent yields, solvent-free conditions, eco-friendly and high catalytic activity make this present procedure an interesting alternative to multistep approach.
ACKNOWLEDGEMENTS
We gratefully acknowledge financial support from the young Researchers and Elite Club Islamic Azad University of Shiraz.
REFERENCES
Ahmad, B., Khan, R.A., Habibullah, A., and Keshai, M. 2009. An improved synthesis of biginelli-type compounds via phase-transfer catalysis. Tetrahydron Letters. 50(24): 2889-2892. https://doi.org/10.1016/j.tetlet.2009.03.177
Alvim, H.G.O., Da Silva Júnior, E.N., and Neto, B.A.D. 2014. What do we know about multicomponent reactions? Mechanisms and trends for the Biginelli, Hantzsch, Mannich, Passerini and Ugi MCRs. RSC Advances. 4(97): 54282-54299. https://doi.org/10.1039/C4RA10651B
Ashok, M., Holla, B.S., and Kumar, N.S. 2007. Convenient one pot synthesis of some novel derivatives of thiazolo[2,3-b]dihydropyrimidinone possessing 4-methylthiophenyl moiety and evaluation of their antibacterial and antifungal activities. European Journal of Medicinal Chemistry. 42(3): 380-385. https://doi.org/10.1016/j.ejmech.2006.09.003
Aswin, K., Mansoor, S.S., Logaiya, K., Sudhan, P.N., and Ahmed, R.N. 2014. Facile synthesis of 3, 4-dihydropyrimidin-2 (1H)-ones and-thiones and indeno [1, 2-d] pyrimidines catalyzed by p-dodecylbenzenesulfonic acid. Journal of Taibah University for Science. 8: 236-247. https://doi.org/ 10.1016/j.jtusci.2014.03.005
Attri, P., Bhatia, R., Gaur, J., Arora, B., Gupta, A., Kumar, N., and C hoi, E.H. 2017. Triethylammonium acetate ionic liquid assisted one-pot synthesis of dihydropyrimidinones and evaluation of their antioxidant and antibacterial activities. Arabian Journal of Chemistry. 10(2): 206-214. https://doi.org/ 10.1016/j.arabjc.2014.05.007
Bewley, C.A., Ray, S., Cohen, F., Collins, S.K., and Overmann, L.E. 2004. Inhibition of HIV-1 envelope-mediated fusion by synthetic batzelladine analogues. Journal of Natural Products. 67(8): 1319-1324. https://doi.org/ 10.1021/np049958o
Biginelli, P. 1893. Aldehyde-urea derivatives of aceto- and oxaloacetic acids. Gazzetta Chimica Italiana. 23(1): 360-413.
Chitra, S., and Pandiarajan, K. 2009. Calcium fluoride: an efficient and reusable catalyst for the synthesis of 3, 4-dihydropyrimidin-2(1H)-ones and their corresponding 2(1H) thione: an improved high yielding protocol for the Biginelli reaction. Tetrahdron Letters. 50(19): 2222-2224. https://doi.org/ 10.1016/j.tetlet.2009.02.162.
De Souza, R., da Penha, E.T., Milagre, H.M.S., Garden, S.J., Esteves, P.M., Eberlin, M.N., and Antunes, O.A.C. 2009. The three‐component Biginelli reaction: a combined experimental and theoretical mechanistic investigation. Chemistry–A European Journal. 15(38): 9799–9804. https://doi.org/10.1002/chem.200900470
Heys, L., Moore, C.G., and Murphy, P. 2000. The guanidine metabolites of Ptilocaulis spiculifer and related compounds; isolation and synthesis. Chemical Society Reviews. 29(1): 57-67. https://doi.org/10.1039/A903712H
Hurst, E.W., and Hull, R. 1961. Two new synthetic substances active against viruses of the psittacosis-lymphogranuloma-trachoma group. Journal of Medicinal and Pharmaceutical Chemistry. 3(2): 215-219. https://doi.org/ 10.1021/jm50015a002
Kamal, A., Krishnaji, T., and Azhar, M.A. 2007. Copper (II) tetrafluoroborate as a mild and efficient catalyst for the one-pot synthesis of 3, 4-dihydropyrimidin-2(1H)-ones under solvent-free conditions. Catalysis Communications. 8(12): 1929-1993. https://doi.org/10.1016/j.catcom.2007.03.009
Kapoor, T.M., Mayer, T.U., Coughlin, M.L., and Mitchison, T.J. 2000. Probing spindle assembly mechanisms with monastrol, a small molecule inhibitor of the mitotic kinesin, Eg5. Journal of Cell Biology. 150(5): 975-988. https://doi.org/10.1083/jcb.150.5.975
Khodja, I.A., Boulcina, R., and Debache, A. 2014. Copper(II) acetate promoted facile synthesis of dihydropyrimidinone derivatives via a solvent free Biginelli multicomponent reaction. Journal of Chemical and Pharmaceutical Research. 6(5): 1040-1045.
Kumar, A., and Maurya, R.A. 2007. An efficient bakers, yeast catalyzed synthesis of 3, 4-dihydropyrimidin-2-(1H)-ones. Tetrahedron Letters. 48(26): 4569-4571. https://doi.org/10.1016/j.tetlet.2007.04.130
Kuraitheerthakumaran, A., Pazhamalai, S., and Gopalakrishnan, M. 2016. Microwave-assisted multicomponent reaction for the synthesis of 3,4-dihydropyrimidin-2(1H)-ones and their corresponding 2(1H)-thiones using lanthanum oxide as a catalyst under solvent-free conditions. Arabian Journal of Chemistry. 9(1): S461-S465. https://doi.org/10.1016/j.arabjc. 2011.06.005
Lal, J., Sharma, M., Gupta, S., Parashar, P., Sahu, P., and Agarwal, D.D. 2012. Hydrotalcite: A novel and reusable solid catalyst for one-pot synthesis of 3, 4-dihydropyrimidinones and mechanistic study under solvent free conditions. Journal of Molecular Catalysis A: Chemical. 352(1): 31-37. https://doi.org/10.1016/j.molcata.2011.09.009
Lashkari, M., Heydari, R., and Mohamadpour, F. 2018. A facile approach for one-pot synthesis of 1H-pyrazolo [1,2-b]phthalazine-5,10-dione derivatives catalyzed by ZrCl4 as an efficient catalyst under solvent-free conditions. Iranian Journal of Science and Technology, Transactions A: Science. 42(3): 1191-1197. https://doi.org/10.1007/s40995-016-0122-8
Litvic, M., Vecani, I., Ladisic, Z.M., Lovric, M., Voncovic, V., Filipan-Litvic, M. 2010. First application of hexaaquaaluminium(III) tetrafluoroborate as a mild, recyclable, non-hygroscopic acid catalyst in organic synthesis: a simple and efficient protocol for the multi gram scale synthesis of 3,4-dihydropyrimidinones by Biginelli reaction. Tetrahedron. 66(19): 3463-3471. https://doi.org/10.1016/j.tet.2010.03.024
Liu, Y.P., Zheng, P., Sun, Z.H., Ye, N., Dong, J.J., and Zhu, L.L. 2008. Economical succinic acid production from cane molasses by Actinobacillus succinogenes. Bioresource Technology. 99(6): 1736-1742. https://doi.org/10.1016/j.biortech.2007.03.044
Magerramow, A.M., Kurbanova, M.M., Abdinbekova, R.T., Rzaeva, I.A., Farzaliev, V.M., and Allokhverdiev, M.A. 2006. Synthesis and antioxidative properties of some 3, 4-dihydropyrimidin-2 (1H) ones (-thiones). Russian Journal of Applied Chemistry. 79(5): 787-790. https://doi.org/10.1134/ S107042720605017X
Mohamadpour, F. 2019. Glutamic acid as green and bio-based α-amino acid catalyst promoted one-pot access to polyfunctionalized dihydro-2-oxypyrroles. Journal of the Serbian Chemical Society. 83(0):1-10 https://doi.org/10.2298/JSC180720006M
Mohamadpour, F. 2018a. Ascorbic acid as a natural green, highly efficient and economical catalyst promoted one-pot facile synthesis of 12-aryltetrahydrobenzo[a]xanthenes-11-ones, 1,8-dioxo octahydroxanthenes and 14-aryl-14H-dibenzo[a,j]xanthenes under solvent-free conditions. UPB Scientific Bulletin, Series B: Chemistry and Materials Science. 80(2): 101-116.
Mohamadpour, F. 2018b. Green and convenient one-pot access to polyfunctionalized piperidine scaffolds via glutamic acid catalyzed knoevenagel-intramolecular [4+2] aza-diels-alder imin-based multi-component reaction under ambient temperature. Polycyclic Aromatic Compounds. https://doi.org/10.1080/10406638.2018.1472111
Mohamadpour, F. 2018c. ZnSO4.7H2O catalyzed one-pot and facile synthesis of highly substituted dihydro-2-oxopyrroles at room temperature. Makara Journal of Science. 22(2): 82-88. https://doi/org/10.7454/mss.v22i2.8792
Mohamadpour, F., Maghsoodlou, M.T., Heydari, R., and Lashkari, M. 2016. Copper (II) acetate monohydrate: An efficient and ecofriendly catalyst for the one-pot multi-component synthesis of biologically active spiropyrans and 1Hpyrazolo[1,2-b]phthalazine-5,10-dione derivatives under solvent-free conditions. Research on Chemical Intermediates. 42(12): 7841-7853. https://doi.org/10.1007/s11164-016-2565-0
Mohamadpour, F., Maghsoodlou, M.T., Heydari, R., and Lashkari, M. 2017. Tartaric acid: A naturally green and efficient di-functional brønsted acid catalyst for the one-pot four-component synthesis of polysubstituted dihydropyrrol-2-ones at ambient temperature. Iranian Journal of Science and Technology, Transactions A: Science. 41(3): 843-849. https://doi.org/ 10.1007/s40995-016-0049-0
Prakash, O., Kumar, R., and Parkash, V. 2008. Synthesis and antifungal activity of some new 3-hydroxy-2-(1-phenyl-3-aryl-4-pyrazolyl) chromones. European Journal of Medicinal Chemistry. 43(2): 435-440. https://doi.org/ 10.1016/j.ejmech.2007.04.004
Rao Jetti, S., Verma, D., and Jain, Sh. 2017. 3-[(3-(Trimethoxysilyl) propyl)thio]propane-1-oxy-sulfonic acid: An efficient recyclable heterogeneous catalyst for the synthesis of 3,4-dihydropyrimidin-2(1H)-ones/thiones. Arabian Journal of Chemistry. 10(2): S3184-S3189. https://doi.org/10.1016/j.arabjc.2013.12.012
Safaei-Ghomi, J., Tavazo, M., and Mahdavinia, G.H. 2018. Ultrasound promoted one-pot synthesis of 3,4-dihydropyrimidin-2(1H)-ones/thiones using dendrimer-attached phosphotungstic acid nanoparticles immobilized on nanosilica. Ultrasonics – Sonochemistry. 40(A): 230–237. https://doi.org/ 10.1016/j.ultsonch.2017.07.015
Slimi, H., Moussaoui, Y., and Salem, R. 2016. Synthesis of 3,4-dihydropyrimidin-2(1H)-ones/thiones via Biginelli reaction promoted by bismuth(III)nitrate or PPh3 without solvent. Arabian Journal of Chemistry. 9(1): S510-S514. https://doi.org/10.1016/j.arabjc.2011.06.010
Shahid, A., Ahmed, N.S., Saleh, T.S., Al-Thabaiti, S.A., Basahel , S.N., Schwieger, W., and Mokhtar, M. 2017. Solvent-free biginelli reactions catalyzed by hierarchical zeolite utilizing a ball mill technique: A green sustainable process. Catalysts. 7(3): 84. https://doi.org/10.3390/ catal7030084
Thompson, J.L., and Hinton, M. 1997. Antibacterial activity of formic and propionic acids in the diet of hens on Salmonellas in the crop. British Poultry Science. 38(1): 59-65. https://doi.org/10.1080/00071669708417941
Wisen, S., Androsavich, J., Evans, C.G., Chang, L., and Gestwi cki, J.E. 2008. Chemical modulators of heat shock protein 70 (Hsp70) by sequential, microwave-accelerated reactions on solid phase. Bioorganic & Medicinal Chemistry Letters. 18(1): 60-65. https://doi.org/10.1016/j.bmcl.2007.11.027
Zhang, Y., Wang, B., Zhang, X., Huang, J., and Liu, C. 2015. An efficient synthesis of 3, 4-dihydropyrimidin-2(1H)-ones and thiones catalyzed by a novel brønsted acidic ionic liquid under solvent-free conditions. Molecules. 20(3): 3811-3820. https://doi.org/10.3390/molecules20033811
Farzaneh Mohamadpour*
Young Researchers and Elite Club, Shiraz Branch, Islamic Azad University, Shiraz, Iran
*Corresponding author. E-mail: mohamadpour.f.7@gmail.com
Total Article Views
Article history:
Received: April 8, 2019
Revised: April 24, 2019
Accepted: May 5, 2019

