
Lactobacillus Enriched Intermediate-Moisture Fruit Products
Tri Indrarini Wirjantoro* , Aphirak Phianmongkhol and Hathaitip RongkomPublished Date : 2019-08-24
DOI : 10.12982/CMUJNS.2015.0078
Journal Issues : Number 2, May - August 2015
ABSTRACT
This study fortified partially-dried apple and cantaloupe with Lactobacillus spp. by employing vacuum impregnation and vacuum drying techniques using impregnation media of sucrose solution and whole milk and two species of Lactobacillus, Lactobacillus acidophilus and Lactobacillus casei. The lightness, firmness and moisture content of the lactobacilli-impregnated fruits were depended on the type of fruit and impregnation solution. The species of Lactobacillus did not produce any significant effect on the fruit microbial property. The final apple and cantaloupe products contained lactobacilli numbers exceeding 109 cfu/g sample. Scanning electron micrographs of the intermediate-moisture apple and cantaloupe clearly demonstrated that the L. acidophilus cells were impregnated in the apple and cantaloupe tissues.
Keywords: Lactobacillus acidophilus, Lactobacillus casei, Intermediate-moisture fruits, Apple, Cantaloupe
INTRODUCTION
The development of functional food products from fruit and vegetables with high number of beneficial microorganism provides an opportunity for the functional food industry, and can also be used to reduce the disadvantages and side effects of milk-based products (Betoret et al., 2012). The interest in fruit products containing probiotics, with their potential health benefits, is growing globally (Lavermicocca, 2006). Lactobacillus acidophilus and Lactobacillus casei are lactic acid bacteria with probiotic capacity, imparting beneficial effects on the host when administered in appropriate amounts (Nighswonger et al., 1996; Chaikham et al., 2013).
Vacuum impregnation is a useful technique to introduce desirable solute into the porous structure of food. The method can conveniently modify the original composition of a food product to develop new products; it has been applied with fruits and vegetables (Zhao and Xie, 2004). According to Chiralt et al. (1999), the application of vacuum impregnation does not disrupt the cellular structure of food, including fruit and vegetable products. Alzamora et al. (2005) fortified apple cylinders with Bifidobacterium spp., Saccharomyces cerevisiae, L. acidophilus and Phoma glomerata using vacuum impregnation. Betoret et al. (2003) applied a similar method to fortify S. cerevisiae and L. casei subsp. rhamnosus in apple cylinders. In both reports, the impregnated apple samples contained around 107 cfu/g of the studied microorganism (Betoret et al., 2003; Alzamora et al., 2005). Vacuum drying conserves and prolongs the shelf life of fruits. Vacuum drying improved the quality of dried fruits compared to conventional drying methods (Wu et al., 2007). Previous studies that dried vacuum-impregnated samples used an air drier (Betoret et al., 2003) or a combination technique of air drying and air microwave (Contreras et al., 2005), whereas our study utilised a vacuum drying method. With vacuum drying, we expect the final product to largely retain the supplemented beneficial organisms, such as probiotics. The purpose of this research was to investigate the survival of two probiotic microorganisms, L. acidophilus and L. casei, in intermediate-moisture apple and cantaloupe pieces by pre-treating with vacuum impregnation and then vacuum drying.
MATERIALS AND METHODS
Microorganisms and their growth
Lyophilised cultures of L. acidophilus TISTR 1338 and L. casei TISTR 1340 were obtained from the Microbiological Resources Center (Bangkok, Thailand). Lactobacilli cells were rehydrated in 5 ml of MRS broth (Merck, Germany) for 10 min at room temperature and anaerobically incubated at 37°C for 24 h. The MRS broth contained 10.0 g/l peptone from casein, 8.0 g/l meat extract, 4.0 g/l yeast extract, 20.0 g/l D(+) glucose, 2.0 g/l di-potassium hydrogen phosphate, 1.0 g/l Tween 80, 2.0 g/l di-ammonium hydrogen citrate, 5.0 g/l sodium acetate, 0.2 g/l magnesium sulfate and 0.04 g/l manganese sulfate. Stock cultures were prepared by mixing 1 ml of young lactobacilli cultures in MRS broth with 1 ml of sterile glycerol in a sterile cryogenic vial and stored at -80°C until used.
Impregnation liquid preparation
Stock cultures were recovered by inoculating 1 ml of the culture into 9 ml of fresh MRS broth (Ashraf and Shah, 2011) and incubated at 37°C for 24 h in an anaerobic environment. On the following day, 5 ml of recovered culture was inoculated into 500 ml of MRS broth and anaerobically incubated at 37°C for 18 h to achieve the beginning of the lactobacilli stationary stage. Following the proliferation of the bacteria that produced a microbial population of approximately 1010 cfu/ml, the cultures were harvested by centrifugation at 1,050 g (3,000 rpm) at 4°C for 20 min (Chan and Zhang, 2002) and washed twice with 0.1% sterilised peptone water (Merck, Germany). The centrifuged culture was then aseptically inoculated into 500 ml impregnation medium, which was either sucrose solution or whole milk (Foremost, Thailand). The aw of the impregnation liquid was previously adjusted to be equalled to the aw of the fresh fruits by adding commercial sucrose (Mitrphol, Thailand). The aw of apple was 0.993 ± 0.010 and cantaloupe 0.992 ± 0.010.
Fresh fruits
Fresh apple (Malus sylvestris Mill var. Granny smith) and cantaloupe (Cucumis melo L. var. cantalupensis) were purchased from a local market and selected according to a similar size and ripeness index (%Brix/acidity) in order to obtain homogeneous samples. On the day of the experiment, the edible portions of the fruits were cut into 1.2×1.2×0.8 cm3.
Production of Lactobacillus enriched intermediate-moisture apple and cantaloupe
A modified method of Betoret et al. (2003) was used for vacuum impregnation. In brief, vacuum impregnation with the impregnation liquid obtained from the above procedure was performed at 25°C in a vacuum oven (Binder VD23, Germany). A vacuum pressure of 50 mbar for 20 min was applied during the vacuum impregnation, followed by a restoration period under the impregnation liquid for 20 min at atmospheric pressure. The vacuum impregnation treatment was carried out using a ratio of 1/5 (w/w) for fruit pieces/impregnation liquid. The fruit pieces were submerged in the impregnation medium throughout the vacuum impregnation process. After vacuum impregnation, the impregnated apple and cantaloupe samples were vacuum dried at a temperature of 58°C at 200 mbar for 9 h and at a temperature of 52°C at 107 mbar for 13 h, respectively, in the vacuum oven.
Determination of physicochemical characteristics
Moisture content of fruit samples was determined according to AOAC methods no. 942.15 (AOAC, 2000). Colour parameter (CIE L* (lightness value)) of the fruit was evaluated by a Minolta colorimeter (Minolta CR-300, Japan). Texture analysis in terms of fruit firmness was analysed using a texture analyzer (XTPL TAXT Plus Texture analyzer, UK) carried out at 25°C. The fruit samples were compressed until 60% strain at a deformation rate of 2 mm/s. The maximum force was recorded as the firmness value of the fruit samples. The firmness of the samples was determined in 10 replications.
Lactobacillus spp. enumeration
Intermediate-moisture fruit samples of 10 g each (Kasımoğlu et al., 2004) were homogenised with 90 ml of sterile Maximum Recovery Diluent (MRD; Difco, USA) using a stomacher for 2 min. Serial dilutions were prepared in MRD and 1 ml of appropriate dilutions were poured in duplicate plates of MRS agar (Merck, Germany), which was adjusted to pH 5.4 (Kenny et al., 2003; Kasımoğlu et al., 2004) by acetic acid (Merck, Germany), and incubated at 37°C for 48 h. Lima et al. (2009) reported that the MRS medium at pH 5.4 was suitable for acidtolerant lactobacilli. Growth colonies of lactobacilli were counted at the end of the incubation time and expressed as logarithmic colony forming units per gram (log cfu/g) of sample.
Microscopic observations
The morphology of dried fruits with probiotic bacteria was examined using a Scanning Electron Microscope (SEM, JEOL JSM-5910LV, Japan) operating at 15 kV accelerating voltage. Apple and cantaloupe tissues were initially fixed in 2.5% glutaraldehyde by immersion. The samples were placed in buffered 2.5% glutaraldehyde and post fixed in buffered 1% osmium tetroxide. The samples were dehydrated by immersion in ethanol solution, and then dried to the critical point in a dryer. The samples were cut across and fixed on the SEM stub, which were subsequently coated with gold in order to provide a reflective surface for the electron beam. The gold-coated samples were subsequently viewed under the microscope (Giri and Suresh, 2007).
Statistical analysis
All data were expressed as mean ± standard deviation of triplicate determinations. The experiment was set up using a Complete Randomized Design with three replications. Analysis of variance (ANOVA-one way) was performed on the experimental results to determine the effect of treatments on different parameters of intermediate moisture fruits. Mean differences and Duncan’s New Multiple Range Test were analysed using SPSS Statistics Base version 17.0 for Windows, serial number 5068035 (SPSS Inc., Chicago, USA).
RESULTS
Physicochemical properties of intermediate-moisture apple and cantaloupe
Several physicochemical properties, including lightness, firmness and moisture content, of partially-dried apple and cantaloupe are displayed in Table 1. Collected data showed that the firmness of fruit impregnated with whole milk was significantly higher than those processed with sucrose solution (P<0.05). The firmness of cantaloupe was lower than apple, particularly after processing the fruit pieces with sucrose solution.
The impregnation solution affected the lightness of the intermediate-moisture fruits. Using whole milk as an impregnation solution produced higher lightness values than using sucrose solution. In addition, the whole milk caused the partially-dried fruit to have significantly higher moisture content than those samples processed with sucrose solution, irrespective of the fruit type (P<0.05). Apple samples had significantly higher moisture content than cantaloupe (P<0.05).
Table 1. Effects of impregnation media and Lactobacillus spp. on the physicochemical properties of impregnated apple and cantaloupe pieces.
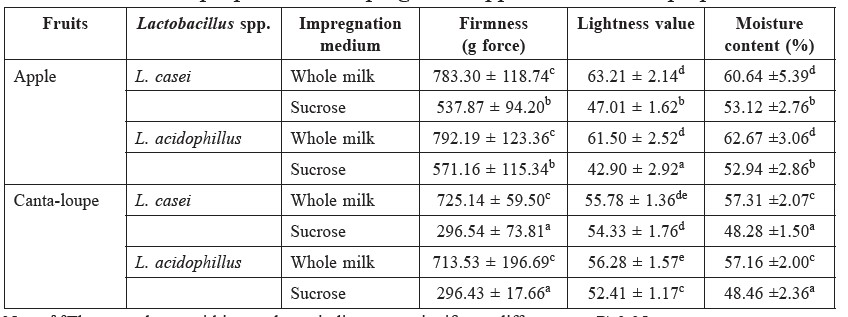
Note: a-eThe same letter within a column indicates no significant difference at P≥0.05.
Enumeration of Lactobacillus spp. in intermediate-moisture apple and cantaloupe
The apple and cantaloupe samples after vacuum impregnation and vacuum drying were assessed for their lactic acid bacteria numbers (Figure 1). The number of lactic acid bacteria in the fresh fruit was less than 0.46 log cfu/g (data not shown). After impregnation, the number of lactic acid bacteria in both of the studied fruits significantly increased to more than 8.03 log cfu/g. This number further increased to reach a maximum of 9.52 log cfu/g after vacuum drying. Different species of Lactobacillus and impregnation solutions did not significantly affect the number of lactobacilli in the intermediate-moisture apple and cantaloupe. Figure 2 displays a microscopic observation of intermediate moisture apple and cantaloupe using SEM.
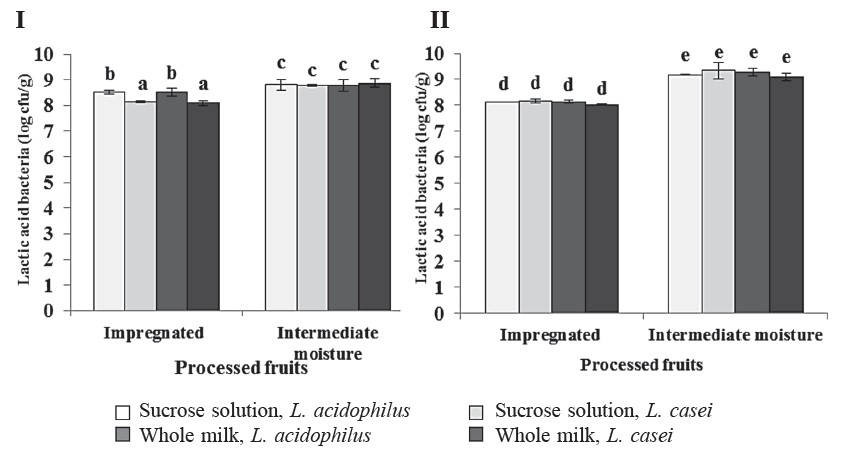
Figure 1. The number of lactic acid bacteria (log cfu/g) in the impregnated and intermediate-moisture apple (I) and cantaloupe (II) enriched with Lactobacillus
spp. using different impregnation media.
DISCUSSION
The physicochemical features of intermediate-moisture fruit, including firmness, lightness and moisture content, were mainly affected by the type of fruit and impregnation liquid. Although the colour values of a* and b* were also assessed, the data displayed a similar trend as the L* value or lightness of the fruit. All of the fruit samples had a water activity between 0.86 and 0.89 (data not shown). Escobedo-Avellaneda et al. (2012) categorised food products with a water activity between 0.6 and 0.9 as intermediate-moisture food.

Figure 2. Lactobacillus acidophillus cells in intercellular space of intermediate apple (A and B) and cantaloupe (C and D) tissue at 3,000x (A and C) and 7,000x (B and D) magnifications.
Impregnation with whole milk produced significantly higher firmness values than sucrose solution (P<0.05). This could be partly due to the larger molecular weight of the milk components compared to sucrose, causing the milk components to impregnate the fruit tissue less and, therefore, produce less damage (Mújica-Paz et al., 2003; Zhao and Xie, 2004). Whole milk also resulted in higher lightness values in the intermediate-moisture apple and cantaloupe than those processed in the sucrose solution. This was because of the lower amount of air in the fruit pores replaced by the whole milk components (Chiralt and Talens, 2005). According to Zhao and Xie (2004), higher gas-liquid exchange in fruit pieces produced a more homogenous refraction index in the sample. The whole milk solution produced significantly higher moisture content compared with the sucrose solution (P<0.05). Lower diffusion of milk components compared to sucrose during vacuum drying could partially explain this result (Wu et al., 2007).
The natural characteristics of each fruit type contributed to the higher firmness and lightness values of intermediate-moisture apple compared to cantaloupe. Fresh apple had a firmness of 2502.38 ± 334.47 g force and a lightness value of 77.68 ± 0.85, while the firmness and lightness values of fresh cantaloupe were 1449.02 ± 125.43 g force and 19.89 ± 1.66, respectively. Although the moisture content of fresh apple (89.00 ± 3.79%) was lower than that of the fresh cantaloupe (93.32 ± 0.13%), the intermediate-moisture apple had higher moisture content than cantaloupe. This finding could be affected by the different vacuum drying conditions of the two fruits.
The vacuum impregnation combined with vacuum drying produced a functional food product; the high number of lactic acid bacteria in the final fruit products in this study was adequate to deliver probiotic effects, exceeding the minimum requirement of 107 cfu/g according to Kourkoutas et al. (2005). The SEM result demonstrated that the cells of L. acidophilus were impregnated in the apple and cantaloupe tissues. This finding confirmed that during impregnation, the whole milk or sucrose solution entered the intercellular spaces of the fruit, together with the lactobacilli cells; this could be due to the pressure gradient during treatment (Betoret et al., 2003).
CONCLUSION
Results of this research strongly demonstrated that apple and cantaloupe show potential to deliver probiotic microorganisms, particularly Lactobacillus spp. The intermediate-moisture apple and cantaloupe contained lactobacilli of more than 9.0 log cfu/g by applying a combination process of vacuum impregnation and vacuum drying. Species type of the Lactobacillus did not significantly affect the final microbial numbers in the intermediate-moisture apple and cantaloupe. However, the selection of impregnation solution and type of fruit affected the firmness, lightness and moisture content of the final products. The electron micrographs confirmed that the lactobacilli cells were impregnated in the intercellular space of apple and cantaloupe.
REFERENCES
Alzamora, S.M., D. Salvatori, M.S. Tapia, A. López-Malo, J. Welti-Chanes, and P. Fito. 2005. Novel functional foods from vegetable matrices impregnated with biologically active compounds. Journal of Food Engineering 67: 205-214.
Ashraf, R., and N.P. Shah. 2011. Selective and differential enumerations of Lactobacillus delbrueckii subsp. bulgaricus, Streptococcus thermophilus, Lactobacillus acidophilus, Lactobacillus casei and Bifidobacterium spp. in yoghurt – A review. International Journal of Food Microbiology 149: 194-208.
Association of Official Analytical Chemists. 2000. Official methods of analysis of AOAC International, 17th ed. AOAC International, Gaithersburg, USA.
Betoret, E., N. Betoret, A. Arilla, M. Bennár, C. Barrera, P. Codoňer, and P. Fito. 2012. No invasive methodology to produce a probiotic low humid apple snack with potential effect against Helicobacter pylori. Journal of Food Engineering 110: 289-293.
Betoret, N., L. Puente, M.J. Díaz, M.J. Pagán, M.J. García, M.L. Gras, J. Martínez-Monzó, and P. Fito. 2003. Development of probiotic-enriched dried fruits by vacuum impregnation. Journal of Food Engineering 56 (2-3): 273-277.
Chan, E.S., and Z. Zhang. 2002. Encapsulation of probiotic bacteria Lactobacillus acidophilus by direct compression. Trans IChemE Journal 80 (Part C): 78-82.
Chaikham, P., A. Apichartsrangkoon, S. Worametrachanon, W. Supraditareporn, E. Chokiatirote, and T. van der Wiele. 2013. Activities of free and encapsulated Lactobacillus acidophilus LA5 or Lactobacillus casei 01 in processed longan juices on exposure to simulated gastrointestinal tract. Journal Science Food Agriculture 93(9): 2229-2238.
Chiralt, A., P. Fito, A. Andres, J.M. Barat, J. Martinez-Monzó, and N. Martinez-Navarrete. 1999. Vacuum impregnation: A tool in minimally processing of foods. p. 314-356. In F.A.R. Oliveira and J.C. Oliveira (eds.) Processing of foods: Quality optimization and process assessment. CRC Press, Boca Raton, USA.
Chiralt, A., and P. Talens. 2005. Physical and chemical changes induced by osmotic dehydration in plant tissues. Journal of Food Engineering 67(1-2): 167-177.
Contreras, C., M.E. Martín, N. Martínez-Navarrete, and A. Chiralt. 2005. Effect of vacuum impregnation and microwave application on structural changes which occurred during air-drying of apple. LWT - Food Science and Technology 38: 471-477.
Escobedo-Avellaneda, Z., G. Velazquez, J.A. Torres, and J. Welti-Chanes. 2012. Inclusion of the variability of model parameters on shelf-life estimations for low and intermediate moisture vegetables. LWT-Food Science and Technology 47: 364-370.
Giri, S.K., and P. Suresh. 2007. Drying kinetics and rehydration characteristics of microwave-vacuum and convective hot-air dried mushrooms. Journal of Food Engineering 78: 512-521.
Kasımoğlu, A., M. Göncüoğlu, and S. Akgün. 2004. Probiotic white cheese with Lactobacillus acidophilus. International Dairy Journal 14: 1067-1073.
Kenny, O., R.J. FitzGerald, G. O’Cuinn, T. Beresford, and K. Jordan. 2003. Growth phase and growth medium effects on the peptidase activities of Lactobacillus helveticus. International Dairy Journal 13: 509-516.
Kourkoutas, Y., V. Xolias, M. Kallis, E. Bezirtzoglou, and M. Kanellaki. 2005. Lactobacillus casei cell immobilization on fruit pieces for probiotic additive, fermented milk and lactic acid production. Process Biochemistry 40: 411-416.
Lavermicocca, P. 2006. Highlights on new food research. Digestive and Liver Disease 38(2): S295-S299.
Lima, K.G.de C., M.F. Kruger, J. Behrens, M.T. Destro, M. Landgraf, and B.D.G. de M. Franco. 2009. Evaluation of culture media for enumeration of Lactobacillus acidophilus, Lactobacillus casei and Bifidobacterium animalis in the presence of Lactobacillus delbrueckii subsp. bulgaricus and Streptococcus thermophilus. LWT – Food Science and Technology 42. 491-495.
Mújica-Paz, H., A. Valdez-Fragoso, A. López-Malo, E. Paloub, and J. Welti-Chanes. 2003. Impregnation properties of some fruits at vacuum pressure. Journal of Food Engineering 56: 307-314.
Nighswonger, B.D., M.M. Brashears, and S.E. Gilliland. 1996. Viability of Lactobacillus acidophilus and Lactobacillus casei in fermented milk products during refrigerated storage. Journal of Dairy Science 72: 212-219.
Wu, L., T. Orikasa, Y. Ogawa, and A. Tagawa. 2007. Vacuum drying characteristics of eggplants. Journal of Food Engineering 83: 422-429.
Zhao, Y., and J. Xie. 2004. Practical applications of vacuum impregnation in fruit and vegetable processing. Trends in Food Science and Technology 15: 434-451.
Tri Indrarini Wirjantoro*, Aphirak Phianmongkhol and Hathaitip Rongkom
Division of Food Science and Technology, Faculty of Agro-Industry, Chiang Mai University, Chiang Mai 50100, Thailand
*Corresponding author. E-mail: triindrarini.w@cmu.ac.th
Total Article Views

