
Microbial Survival and Sensory Properties of Intermediate-Moisture Apple and Cantaloupe Impregnated with Lactobacillus acidophilus during Storage
Hathaitip Rongkom, Aphirak Phianmongkhol and Tri Indrarini Wirjantoro*Published Date : 2019-08-24
DOI : 10.12982/CMUJNS.2015.0076
Journal Issues : Number 2, May - August 2015
ABSTRACT
The sensory properties and bacterial viability of intermediate-moisture apple and cantaloupe supplemented with Lactobacillus acidophilus and packed in vacuum packaging were monitored during eight weeks of storage at refrigerated and room temperatures. The intermediate-moisture apple and cantaloupe had higher lightness value and better bacterial survival when the fruit samples were stored at refrigerated temperatures. The numbers of total microorganisms and lactic acid bacteria in the fruit samples reduced significantly to below 1.0 log cfu/g after eight weeks of storage at room temperature. On the other hand, refrigerated storage maintained high numbers of lactic acid bacteria – 7.94±0.02 and 9.44±0.38 log cfu/g, in the apple and cantaloupe samples, respectively, at the end of the storage period. A sensory panel preferred the intermediate-moisture fruits stored at chilled temperatures compared to room temperatures.
Keywords: Lactobacillus acidophilus, apple, cantaloupe, microbial viability, storage period
INTRODUCTION
Development of functional food products is driven by the market potential for food and beverage products that can improve the health and well being of consumers (Prado et al., 2008). These food products are recognised to contain a component (which may or may not be a nutrient) that affects one or a limited number of functions in the body in a targeted way so as to have positive effects on health (Roberfroid, 2000). The presence of probiotics and prebiotics in food products creates functional food products (Mattila-Sandholm et al., 2002). In supplementing food with probiotics, the safety, sensory properties and viability of probiotics during storage must be considered (Saarela et al., 2000). Maintaining probiotic viability at functional levels during storage is a challenge in the production of probiotic functional food products. Aureli et al. (2010) reported that 87% of the commercial probiotic food supplements in Italy did not conform to the Italian guidelines and the differences were both quantitative and qualitative (number determination, purity, types and viability of microorganisms). The results of Gueimonde et al. (2004) for 10 commercial fermented milks in Spain showed nearly similar results during refrigerated storage, with counts of Lactobacillus spp. always higher than 105cfu/ml, whereas the population of Bifidobacterium spp. decreased below this level in two of seven studied products. Kourkoutas et al. (2005) stated that the number of live bacteria in probiotic products should be at least 107cfu/g to achieve health benefits.
Lactobacillus acidophilus is one of the Lactobacillus spp. that has been widely reported to possess probiotic characteristics (Mattila-Sandholm et al., 2002; Prado et al., 2008). Although the bacteria have been applied in several dairy products, such as yoghurt and cheese (Paseephol and Sherkat, 2009; Gomes et al., 2011), a review by Prado et al. (2008) showed that the microorganism could be supplemented in some beverage products, including tomato juice, beet juice and beetroot and carrot juice. For fruit products, Ding and Shah (2008) supplemented orange and apple juice with L. acidophilus, while Ankolekar et al. (2012) utilised L. acidophilus in the fermentation of pear juice. However, no published research has directly supplemented L. acidophilus in fruit pieces and monitored their survival in the products.
Intermediate-moisture food products have a moisture content in the range of 10 to 40%, with water activity between 0.6 and 0.9 (Escobedo-Avellaneda et al., 2012). The lower water activity of the food products, which partly inhibits the growth of microorganisms, increased their shelf life under ambient conditions for up to several months (Taoukis and Richardson, 2007; Sexena et al., 2009; Escobedo-Avellaneda et al., 2012). Intermediate-moisture fruit products retained some nutritional values with less transportation and storage costs compared to the fresh form (Escobedo-Avellaneda et al., 2012). Since the intermediate-moisture food products had plasticity, soft texture and chewability without oral sensation of dryness, the products were suitable for direct consumption without any preparation effort. In addition, the texture of the food products was much closer to normal food than the crisp or rigid structures of fully dehydrated food products (Brockmann, 1970; Taoukis and Richardson, 2007). This research monitored the survival of lactic acid bacteria, in particular L. acidophilus, added to intermediate-moisture apple and cantaloupe during storage at refrigerated and room temperatures, as well as changes in the sensory characteristics of the stored fruits.
MATERIALS AND METHODS
Lactobacillus strain
Lyophilised culture of L. acidophilus TISTR 1338 was obtained from the Microbiological Resources Center (Bangkok, Thailand). The cultures were stored in MRS broth containing 50% glycerol at -80°C before use.
Impregnation liquid preparation
Fresh cultures of L. acidophilus TISTR 1338 were obtained by growing 1 ml of stock culture in 9 ml MRS broth (Merck, Germany) at 37°C for 24 h. On the following day, 5 ml of the fresh culture was inoculated into 500 ml fresh MRS broth and incubated at the same incubation temperature as fresh cultures for 18 h to obtain young cultures of lactobacilli at their maximum concentration. From the last cultivation step, the number of L. acidophilus reached a count of 10 log cfu/ml. To remove the microbiological medium from the bacterial cells, the microbial cells were harvested by centrifugation at 3,000 rpm for 20 min at 4°C (Chan and Zhang, 2002) and then washed twice with 0.1% (w/v) sterilized peptone water (Merck, Germany). The harvested cells were aseptically inoculated into sterile whole milk (Foremost, Thailand), which was previously adjusted with commercial sucrose (Mitrphol, Thailand) to have a water activity similar to the studied fruits. The aw of apple was 0.993 ± 0.010, while cantaloupe had an aw of 0.992 ± 0.010. The L. acidophilus concentration in the impregnation liquid of whole milk was 10 log cfu/ml.
Production and storage of L. acidophilus added intermediate-moisture apple and cantaloupe
Fresh apple (Malussylvestris Mill var. Granny smith) and cantaloupe (Cucumismelo L. var. cantalupensis) were purchased from a local Thai market in Chiang Mai. On the day of the experiment, the edible portions of the fruits were cut into 1.2×1.2×0.8 cm3 pieces. The fruit pieces were submerged in the wholemilk solution containing L. acidophilus at a ratio of 1/5 (w/w) for fruit pieces/impregnation liquid for probiotic impregnation. The impregnation was done at a low vacuum pressure (vacuum impregnation) according to Betoret et al. (2003) with some modifications. In this study, the impregnation treatment was carried out at 25°C in a vacuum oven (Binder VD23, Germany). A vacuum pressure of 50 mbar for 20 min was applied during the vacuum impregnation, followed by a restoration period under the impregnation liquid for 20 min at atmospheric pressure. After the impregnation process, fruit samples were vacuum dried at a temperature of 58°C at 200 mbar for 9 h for apple samples and at a temperature of 52°C at 107 mbar for 13 h for cantaloupe pieces in a vacuum oven. The intermediate-moisture apple and cantaloupe were vacuum-sealed in laminated bags of Polyethylene tetraphthalate / Polypropylene / Aluminum. The fruit samples were then stored at room temperature (30 ± 0.5°C) or in a refrigerator (4 ± 1°C) for eight weeks. During storage, representative samples were analysed every two weeks to monitor changes in the physical and microbial properties of the intermediate-moisture fruits.
Determination of the lightness of intermediate-moisture apple and cantaloupe
The color parameter (CIE L* (lightness value)) of intermediate-moisture fruits was evaluated by a Minolta colorimeter (Minolta CR-300, Japan).
Enumeration of viable cells in the intermediate-moisture apple and cantaloupe
Intermediate moisture fruits (10 g) were homogenised with 90 ml of sterile Maximum Recovery Diluent (MRD) (Difco, USA) using a stomacher for 2 min. Serial dilutions were prepared in MRD and 1 ml of appropriate dilutions were poured in duplicate plates of Plate Count Agar (Merck, Germany) for viable counts of aerobic mesophilic bacteria and MRS agar (Merck, Germany), which was adjusted to pH 5.4 by 5% acetic acid (Merck, Germany), for lactic acid bacteria. Cultures were incubated at 37°C for 48 h. The growth colonies were counted at the end of the incubation time and expressed as logarithmic colony forming units per gram (log cfu/g) of sample.
Sensory evaluation
The sensory quality of the intermediate-moisture fruit pieces were evaluated using a 9-point hedonic scale (ranking from 9-like extremely to 1-dislike extremely) by 50 panelists – undergraduate and postgraduate students of the Division of Food Science and Technology, Faculty of Agro-Industry, Chiang Mai University. Sensory preferences of color, odor, firmness and overall acceptability of the fruit samples were evaluated (Walkling-Ribeiro et al., 2009).
Statistical analysis
The experiment was set up using a Complete Randomized Design with three replications. Analysis of variance (ANOVA-one way) was performed on the experimental results to determine the effect of storage temperatures and storage times on the lightness and microbial numbers of the studied fruits. The sensory evaluation data were statistically analysed using ANOVA by applying a Random Complete Block Design experiment following the method of Sriwimon and Boonsupthip (2011). Mean differences and Duncan’s New Multiple Range Test were analysed using SPSS Statistics Base version 17.0 for Windows, serial number 5068035 (SPSS Inc., Chicago, USA).
RESULTS
Lightness of intermediate-moisture apple and cantaloupe
Visual appearance of fresh-cut fruits is an important parameter of consumer preference (Toivonen and Brummell, 2008). Given this, the color value (represented by lightness) of intermediate-moisture apple and cantaloupe was monitored during storage (Figure 1). Data showed that storing the intermediate-moisture fruits at a refrigerated temperature maintained the lightness of the fruit samples, whereas a slight reduction in the lightness value was observed when the fruit samples were stored at room temperature. The intermediate-moisture cantaloupe and apple had lightness values of 55.07 ± 1.73 and 56.03 ± 1.56, respectively, at the beginning of storage. These values reduced to 51.05 ± 0.73 and 47.11 ± 3.12 in the cantaloupe samples after eight weeks of storage at chilled and room temperatures, respectively, whereas the lightness of apple samples were 55.02 ± 2.39 and 53.53 ± 2.88, respectively (Figure 1).
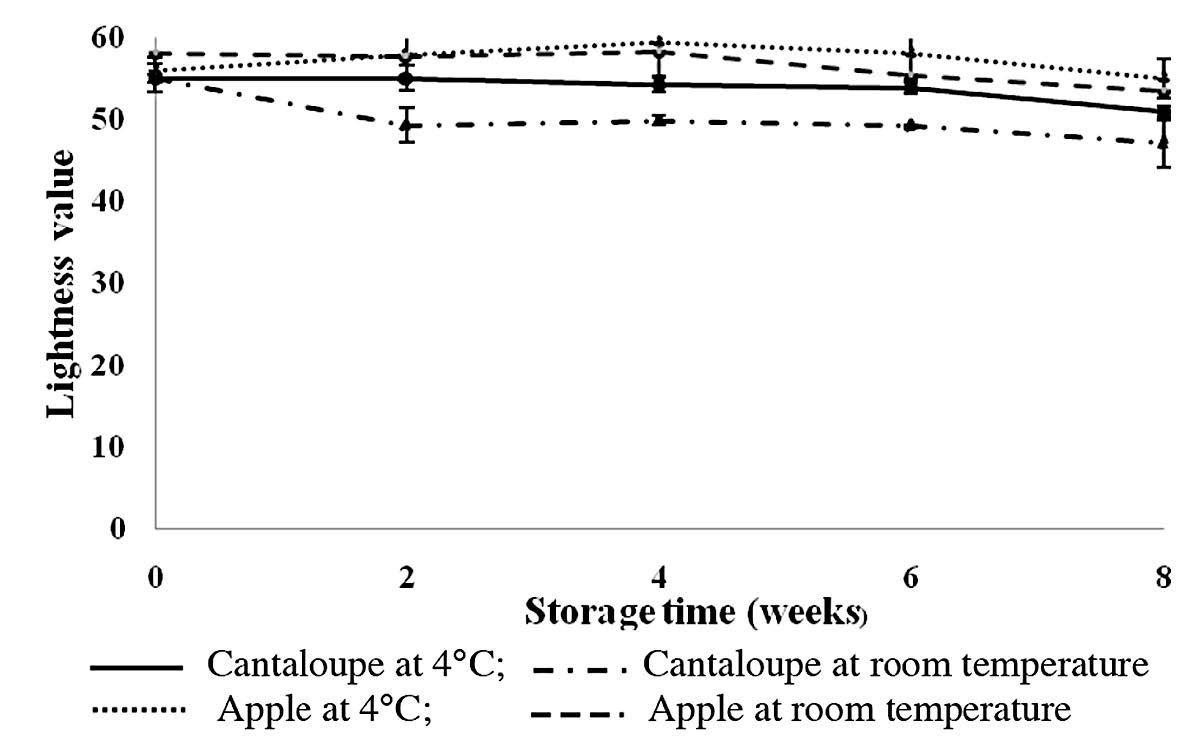
Figure 1. Lightness value of intermediate-moisture apple and cantaloupe supplemented with L. acidophilus during storage at 4°C and room temperature for eight weeks.
Microbial enumeration of intermediate-moisture apple and cantaloupe
Lactic acid bacteria were the main microorganisms in the fruit samples (Figure 2). The survival of these bacteria in the fruit samples during storage depended significantly on the storage temperatures and times. The number of lactic acid bacteria in the apple samples reduced substantially to around 3 log cfu/g after two weeks storage at room temperature, followed by a continuous decline until they were below the detection limit of the pour-plate method (< 10 cfu/g) after six weeks of storage. For cantaloupe, lactic acid bacteria survived at room temperature at 8.48±0.1 log cfu/g after two weeks of storage, but then declined sharply until below the detection limit of the pour-plate method at the end of storage.
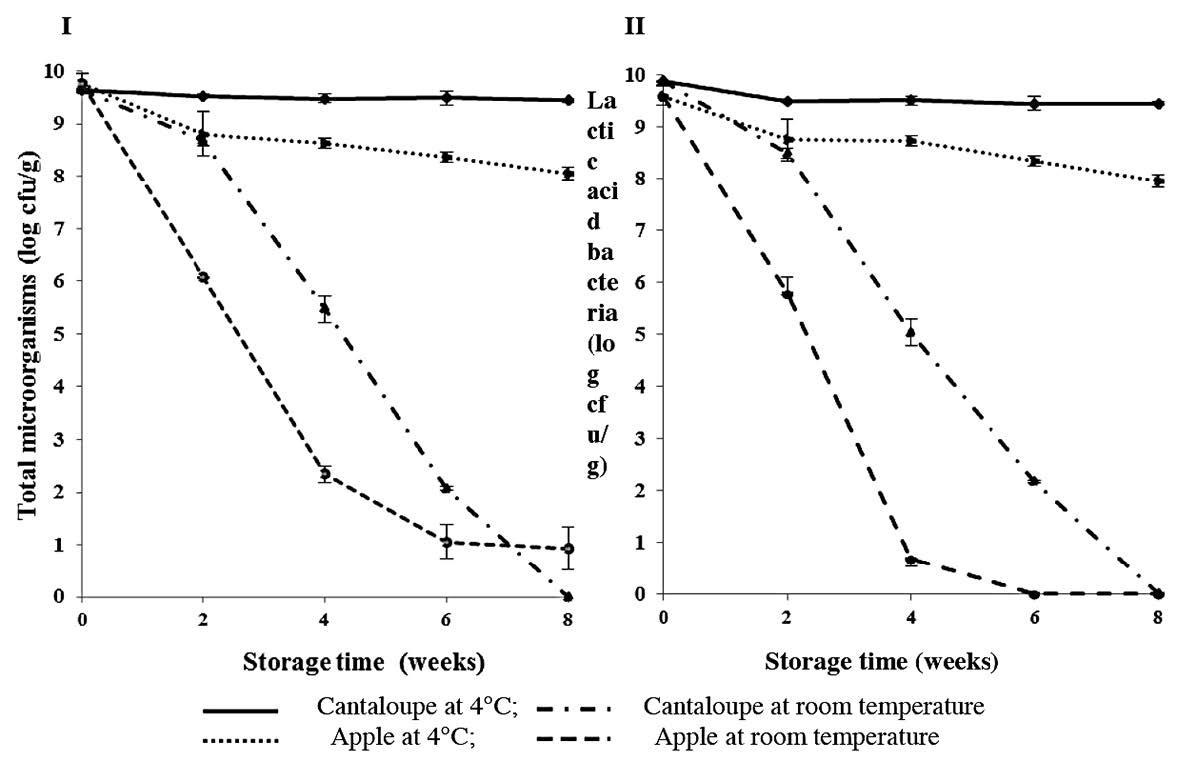
Figure 2. Enumeration of total microorganisms (I) and lactic acid bacteria (II) (log cfu/g) in the intermediate-moisture apple and cantaloupe supplemented with L. acidophilus during storage at 4°C and room temperature for eight weeks.
Table 1. Preference scores of the intermediate-moisture apple and cantaloupe supplemented with L. acidophilus during storage at 4°C and room temperature for eight weeks.
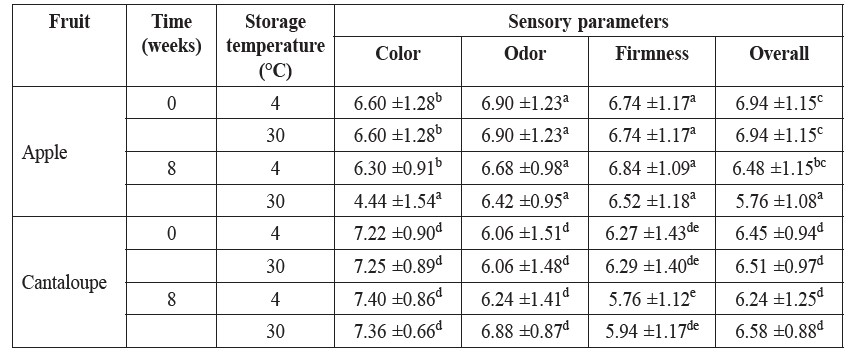
Note: a-d Different letters within the same column and similar fruit type were significantly different by Duncan’s new multiple range test (P<0.05).
Sensory assessment of intermediate-moisture apple and cantaloupe
Data in Table 1 displayed the preference scores of sensory panelists for the intermediate-moisture apple and cantaloupe impregnated with L. acidophilus at the beginning and end of storage. The color, odor and overall acceptability of apple and cantaloupe were maintained by storage at low temperature. However, a significant decrease in the color and overall preferences of apple was perceived when the apple pieces were kept at room temperature instead for eight weeks (P<0.05).
DISCUSSION
Changing of the fruit lightness at higher storage temperature could be due to enzymatic browning that occurred more easily at high temperature (Perez-Cabrara et al., 2011). The lightness finding at room temperature in this study was similar to the report of Raybaudi-Massilia et al. (2007) for Fuji apple. Higher reductions in the lightness value of cantaloupe compared to apple could be affected by an initially higher pH value of the fruit (pH of cantaloupe was 4.11 ± 0.11, while apple was 3.66 ± 0.00). Gomes et al. (2014) reported that plant polyphenoloxidase, which catalysed enzymatic browning, were nearly inactive at pHs lower than 4.0. The results of microbial enumeration in the intermediate-moisture apple and cantaloupe indicated that cantaloupe tissue better protected probiotic bacteria compared to that of apple during storage at chilled and room temperatures. The good result of cantaloupe was similar to the finding of Betoret et al. (2003) that reported impregnated dried apple contained approximately 107 cfu/g Lactobacillus casei subsp. rhamnosus after 15 days storage at 20ºC. The low viability of lactic acid bacteria, mainly L. acidophilus, during the storage period could be affected by the high aw of the fruit samples. The intermediate-moisture apple and cantaloupe stored at room temperature had water activities of 0.87 ± 0.00 and 0.86 ± 0.00, respectively, at the beginning of storage. Makinen et al. (2012) suggested that dehydrated food products, such as cereals, biscuits, snacks and confectionary, should have aw between 0.2 and 0.5 to provide a more favorable condition for probiotic stability.
The survival of lactic acid bacteria in the intermediate-moisture apple and cantaloupe was still high when the fruit samples were kept at the low storage temperature of 4ºC. The decline in the lactic acid bacteria number was less than 1.7 log cfu/g, producing final lactic acid bacteria numbers of 7.94 ± 0.02 and 9.44 ± 0.38 for apple and cantaloupe, respectively. These final levels were higher than the therapeutic number of 107cfu/g for probiotics in dairy products (Betoret et al., 2003). These results agreed with the report of Krasaekoopt and Suthanwong (2008) that reported L.casei01 in impregnated guava and papaya of approximately 107 cfu/g after four weeks storage at refrigerated temperature. A study by Rößle et al. (2010) of apple wedges enriched with Lactobacillus rhamnosus GG showed that the level of probiotic was maintained at approximately 108cfu/g over 10 days at 2-4ºC.
A maximum limit of 1-103cfu/g for moulds and yeasts in fruit products is recommended (Moreno et al., 2012). The intermediate-moisture apple and cantaloupe in this study had moulds and yeasts of less than 1.48 ± 0.18 and 1.23 ± 0.21 log cfu/g when the samples were kept at room and chilled storage temperatures, respectively (data not shown). The low number of moulds and yeasts in this study could be affected by the antimicrobial effectiveness of lactic acid bacteria for nutrient competition and production of antagonistic compounds. Dalié et al. (2010) reviewed that probiotics, including L. acidophilus, were able to synthesize bacteriocins that could inactivate Gram-negative bacteria and eukaryotic microorganisms, such as moulds and yeasts.
A lower preference of the sensory panelists for the intermediate-moisture apple kept at room temperature could be affected by the reduction of the lightness value (Figure 1) due to enzymatic browning. Raybaudi-Massilia et al. (2007) also reported a better acceptance for color of fresh-cut Fuji apples at day 0 compared to those that had been stored for 15 days at 5ºC.
CONCLUSION
Results from this study clearly showed that apple and cantaloupe could be utilised as a good vehicle to deliver probiotic microorganisms to health-conscious consumers. A low storage temperature increased the survival of lactic acid bacteria in the intermediate-moisture apple and cantaloupe. However, the possibility of
further reducing the aw of the intermediate-moisture apple and cantaloupe should not be ruled out to produce a product that would be stable at room temperature for more convenient handling.
REFERENCES
Ankolekar, C., M. Pinto, D. Greene, and K. Shetty. 2012. In vitro bioassay based screening of antihyperglycemia and antihypertensive activities of Lactobacillus acidophilus fermented pear juice. Innovative Food Science and Emerging Technologies 13: 221-230.
Aureli, P., A. Fiore, C. Scalfaro, M. Casale, and G. Franciosa. 2010. National survey outcomes on commercial probiotic food supplements in Italy. International Journal of Food Microbiology 137: 265-273.
Betoret, N., L. Puente, M.J. Díaz, M.J. Pagán, M.J. García, M.L. Gras, J. Martínez-Monzó, and P. Fito. 2003. Development of probiotic-enriched dried fruits by vacuum impregnation. Journal of Food Engineering 56 (2-3): 273-277.
Brockmann, M.C. 1970. Development of intermediate-moisture foods for military use. Food Technology 2(8): 60-64.
Chan, E.S., and Z. Zhang. 2002. Encapsulation of probiotic bacteria Lactobacillus acidophilus by direct compression. Trans IChemE Journal 80 (Part C): 78-82.
Escobedo-Avellaneda, Z., G. Velazquez, J.A. Torres, and J. Welti-Chanes. 2012. Inclusion of the variability of model parameters on shelf-life estimations for low and intermediate-moisture vegetables. LWT-Food Science and Technology 47: 364-370.
Dalié, D.K.D., A.M. Deschamps, and F. Richard-Forget. 2010. Lactic acid bacteria potential for control of mold growth and mycotoxins: A review. Food Control 21: 370-380.
Ding, W.K., and N.P. Shah. 2008. Survival of free and microencapsulated probiotic bacteria in orange and apple juices. International Food Research Journal 15(2): 219-232.
Gomes, A.A., S.P. Braga, A.G. Cruz, R.S. Cadena, P.C.B. Lollo, C. Carvalho, J. Amaya-Farfán, J.A.F. Faria, and H.M.A. Bolini. 2011. Effect of the inoculation level of Lactobacillus acidophilus in probiotic cheese on the physicochemical features and sensory performance compared with commercial cheese. Journal of Dairy Science 94: 4777-4786.
Gomes, M.H., T. Vieira, J.F. Fundo, and D.P.F. Almeida. 2014. Polyphenoloxidase activity and browning in fresh cut ‘Rocha’ pear as affected by pH, phenolic substrates, and antibrowning additives. Postharvest Biology and Technology 91: 32-38.
Gueimonde, M., S. Delgado, B. Mayo, P. Ruas-Madiedo, A. Margolles, and C.G. de los Reyes-Gavilán. 2004. Viability and diversity of probiotic Lactobacillus and Bifidobacterium populations included in commercial fermented milks. Food Research International 37: 839-850.
Kourkoutas, Y., V. Xolias, M. Kallis, E. Bezirtzoglou, and M. Kanellaki. 2005. Lactobacilluscasei cell immobilization on fruit pieces for probiotic additive, fermented milk and lactic acid production. Process Biochemistry 40: 411- 416.
Krasaekoopt, W., and B. Suthanwong. 2008. Vacuum impregnation of probiotics in fruit pieces and their survival during refrigerated storage. Kasetsart Journal 42: 723-731.
Makinen, K., B. Berger, R. Bel-Rhlid, and E. Ananta. 2012. Science and technology for the mastership of probiotic applications in food products. Journal of Biotechnology 162: 356-365.
Mattila-Sandholm, T., P. Myllärinen, R. Crittenden, G. Mogensen, R. Fondén, and M. Saarela. 2002. Technological challenges for future probiotic foods. International Dairy Journal 12: 173-182.
Moreno, J., R. Simpson, N. Pizarro, K. Parada, N. Pinilla, J.E. Reyes, and S. Almonacid. 2012. Effect of ohmic heating and vacuum impregnation on the quality and microbial stability of osmotically dehydrated strawberries (cv. Camarosa). Journal of Food Engineering 110: 310-316.
Paseephol, T., and F. Sherkat. 2009. Probiotic stability of yoghurts containing Jerusalem artichoke inulins during refrigerated storage. Journal of Functional Foods 1: 311-318.
Perez-Cabrera, L., M. Chafer, A. Chiralt, and C. Gonzalez-Martinez. 2011. Effectiveness of anti-browning agents applied by vacuum impregnation on minimally processed pear. Food Science and Technology 44: 2273-2280.
Prado, F.C., J.L. Parada, A. Pandey, and C.R. Soccol. 2008. Review. Trends in non-dairy probiotic beverages. Food Research International 41: 111-123.
Raybaudi-Massilia, R.M., J. Mosqueda-Melgar, A. Sobrino-López, R. Soliva-Fortuny, and O. Martín-Belloso. 2007. Shelf-life extension of fresh-cut “Fuji” apples at different ripeness stages using natural substances. Postharvest Biology and Technology 45: 265-275.
Roberfroid, M.B. 2000. Prebiotics and probiotics: Are they functional foods? The American Journal of Clinical Nutrition 71(suppl): 1682S-1687S.
Rößle, C., M.A.E. Auty, N. Brunton, R.T. Gormley, and F. Butler. 2010. Evaluation of fresh-cut apple slices enriched with probiotic bacteria. Innovative Food Science and Emerging Technologies 11: 203-209.
Saarela, M., G. Mogensen, R. Fondén, J. Mättö, and T. Mattila-Sandholm. 2000. Probiotic bacteria: Safety, functional and technological properties Review: Journal of Biotechnology 84: 197-215.
Sriwimon, W., and W. Boonsupthip. 2011. Utilization of partially ripe mangoes for freezing preservation by impregnation of mango juice and sugars. Food Science and Technology 44(2): 375-383.
Taoukis, P.S., and M. Richardson. 2007. Principles of intermediate-moisture foods and related technologies. p. 273-312. In G.V. Barbosa-Cánovas, A.J. Fontana, Jr., S.J. Schmidt, and T.P. Labuza (eds) Water activity in foods: Fundamentals and applications. Blackwell Publishing and Institute of Food Technologists, Iowa, USA.
Toivonen, P.M.A., and D.A. Brummell. 2008. Review. Biochemical bases of appearance and texture changes in fresh-cut fruit and vegetables. Postharvest Biology and Technology 48: 1-14.
Sexena, S., B.B. Mishra, R. Chander, and A. Sharma. 2009. Shelf stable intermediate-moisture pineapple (Ananascomosus) slices using hurdle technology. LWT - Food Science and Technology 42: 1681-1687.
Walkling-Ribeiro, M., F. Noci, D.A. Cronin, J.G. Lyng, and D.J. Morgan. 2009. Shelf life and sensory evaluation of orange juice after exposure to thermosonication
and pulsed electric fields. Food and Bioproducts Processing 87: 102-107.
Hathaitip Rongkom, Aphirak Phianmongkhol and Tri Indrarini Wirjantoro*
Division of Food Science and Technology, Faculty of Agro-Industry, Chiang Mai University, Chiang Mai 50100, Thailand
*Corresponding author. E-mail: triindrarini.w@cmu.ac.th
Total Article Views

