
Enhancement of Glycyrrhizic Acid and Microbial Decontamination of Glycyrrhiza glabra (licorice) by Gamma Radition
Wachiraporn Pewlong*, Surasak Sajjabut, Jaruratana Eamsiri, Sirilak Chookaew and Kanokporn BoonsirichaiPublished Date : 2019-08-23
DOI : 10.12982/cmujns.2016.00016
Journal Issues : Number3 ,September - December 2016
ABSTRACT
Gamma irradiation is an effective means for controlling pathogenic microorganisms in many herbal materials. This study evaluated the effect of gamma radiation on microbial load, antioxidant activity, and total phenolic content, including the amount of glycyrrhizic acid of Glycyrrhiza glabra, or licorice powder. Gamma irradiation was applied up to 20 kGy (at 5 kGy intervals) to powdered root samples. Total bacteria, yeast and mold, and coliform bacteria were counted after gamma irradiation; all decreased with increasing dose. After irradiation at 5 kGy, total bacterial count and total yeast and mold diminished by 2 log cycles. A dose of 5 kGy was sufficient to eliminate the pathogenic bacteria, such as coliform bacteria Escherichia coli and Clostridium perfringens. The antioxidant properties and total phenolic content did not differ significantly between non-irradiated and irradiated samples. However, treatment with gamma radiation altered the quantity of glycyrrhizic acid; the measured glycyrrhizic acid content increased in samples treated with 15 and 20 kGy radiation. To conclude, the microbial quality of licorice powder can improved by treating with gamma radiation without affecting the antioxidant activity and phenolic compositions. In addition, a gamma irradiation dose higher than 15 kGy enhanced the glycyrrhizic acid content in the extract.
Keywords: Glycyrrhiza glaba, Gamma radiation, Decontamination
INTRODUCTION
Glycyrrhiza glabra, commonly known as licorice, is a Thai traditional medicine belonging to the Fabaceae family. The plant is widely distributed in the subtropical and warm temperature regions of the world. Licorice has a distinctive sweet taste because of glycyrrhizin, which is 50 times sweeter than sugar (Khattak and Simpson, 2010). In traditional medicine, this plant has been used for centuries in the treatment of sore throats, coughs, asthma, gastric ulcers, and allergic reactions. The roots of G. glabra are also used in the tobacco industry to flavor cigarettes, pipe tobacco, and anti-smoking preparations (Khattak and Simpson, 2010). Nowadays, licorice is widely used for its antimicrobial, antioxidant, anti-inflammatory, anticoagulative, antiulcer, antiallergic, and anxiolytic activities (Vaya et al., 1997; Shin et al., 2007). Previous studies have shown that G. glabra consists of major bioactive components such as glycyrrhizin, glycyrrhizic acid, and glabarin A and B (Khattak and Simpson, 2010), as well as other several active chemical substances, including chalones, saponins, flavonoids, isoflavones, coumarins, and triterpenes (Kitakawa, 2002).
Contamination caused by microorganisms and insects remains a challenge for the long-term storage of licorice powder. In food safety, the use of radiation, such as gamma rays, can provide an effective method to eliminate insect and microorganisms to extend the shelf life of various food, fruits, and herbs (Mustapha et al., 2014). Gamma irradiation as a phytosanitary treatment has been proven safe and to effectively improve the hygienic quality of various food and herbal materials (Fargus, 1998). Several researchers have reported microbial contamination in the root of G. glabra (Al-Bachir and Lahham, 2003; Al-Bachir and Zeinou, 2005; Khattak and Simpson, 2010). These reports also revealed that gamma radiation treatment significantly diminished the microbial load in G. glabra root. Nevertheless, the effect of gamma irradiation on the free radical scavenging activity and glycyrrhizic acid content of G. glabra roots has not been investigated. Consequently, we must determine the chemical and physical properties of irradiated products that might be affected by gamma radiation.
MATERIALS AND METHODS
Preparation of licorice extract and gamma irradiation
Licorice powder was brought from a local market in Bangkok, Thailand. One hundred grams of licorice powder was packed in aluminum foil bags and exposed to gamma radiation at doses of 5, 10, 15, and 20 kGy at Gems Irradiation Center, Thailand Institute of Nuclear Technology. Non-irradiated powder served as the control. Ten grams of samples were extracted in 100 mL of 70% methanol for 1 hour in an ultrasonic bath. The extracts were filtered through No. 4 Whatman filter paper.
Microbial load analyses
Microbiological evaluations of irradiated and non-irradiated licorice were conducted or enumerated according to the Association of Official Analytical Chemists (AOAC, 1990). The initial microflora in licorice were determined by homogenizing 25 g of each sample with 225 mL of Butterfield’s phosphate buffer using a lab-blender to give 0.1 dilution (1:10). Serial dilutions up to 10-6 were prepared. For determination of total viable bacterial count and total yeast and mold, 1 mL of appropriate dilutions were assayed by poured plated technique on plate count agar (PCA) and potato dextrose agar (PDA), respectively. All samples were then incubated at 35°C for 48 h and 25°C for 5 days, respectively. The pathogens, such as Coliforms bacteria, Escherichia coli, Bacillus cereus, Clostridium perfringens, and Salmonella spp., were determined according to AOAC (1990).
Free radical-scavenging assay with DPPH and ferric reducing antioxidant potential (FRAP)
The antioxidant activity assay was performed as previously described (Khattak et al., 2008) with slight modifications, in which 1,1-diphenyl-2-picrylhydrazyl (DPPH) was used as a radical scavenger. The free radical-scavenging activity was expressed as ascorbic acid equivalent. FRAP assay was performed according to the method described by Benzei and Strain (Benzei and Strain, 1996). The antioxidant potential of the samples was analyzed based on a calibration curve plotted using FeSO4.7H2O at concentrations ranging from 400 to 2,000 μM.
Total phenolic content
The content of reducing components was estimated using the Folin-Ciocateau assay according to the method developed by Velioglu et al. (1998). The total phenolic content was expressed as gallic acid equivalent (GAE) mg/g.
Glycyrrhizic acid content
The amount of glycyrrhizic acid was performed by HPLC (Jasco, Japan) equipped with a UV detector. The separation was carried out using a C18, Phenomenex column. The glycyrrhizic acid peak was detected at 254 nm. The entire chromatographic separation was performed at an isocratic mobile phase of methanol and 1% (v/v) acetic acid at the ratio 70:30 v/v (Charpe and Rathod, 2015).
Statistical analysis
All measurements were done in triplicate. Results were expressed as mean ± standard deviation (SD). Data were analyzed using one-way ANOVA and DUNCAN tests (SPSS software version 21) for differences between the control and the irradiated samples for all parameters.
RESULTS
Effect of irradiation on the microbial load
The principal aim of irradiation is to totally eradicate microorganisms. The results of total viable bacterial counts indicated that the non-irradiated samples of licorice were highly contaminated with bacteria at the level of 2.67x106 cfu/g, exceeding the maximum permissible limit limit of 1.0x104 cfu/g specified by the World Health Organization (1998). The effects of gamma irradiation at various doses on total viable count and total yeast and mold are shown in Table 1. The initial total bacterial count of non-irradiated samples was 2.67x106 cfu/g, whereas that of samples irradiated at 5 kGy was 2.14x104 cfu/g. Total yeast and mold were eliminated after being irradiated at 5 kGy. Irradiation at 10 kGy was sufficient to practically eliminate bacteria, yeast, and mold.
Table 1. Effects of gamma irradiation on pathogenic microorganisms in licorice powder at varying doses.
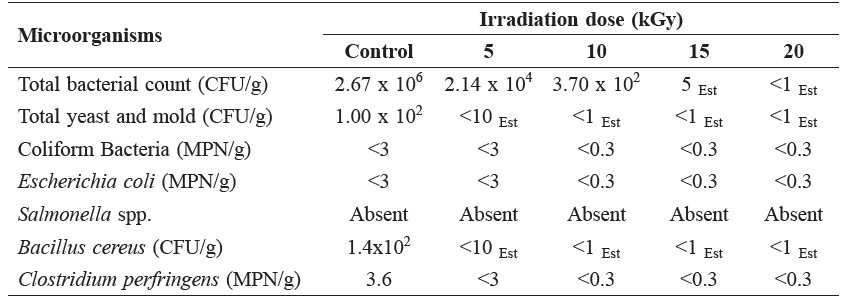
Note: Est = estimated value
Antioxidant activity
Using DPPH and FRAP assays, the antioxidant activities of licorice were determined (Table 2). Gamma irradiation did not show any significant effect on antioxidant activity of the control and irradiated samples. The DPPH activities of the control (9.00 ± 1.12 mgAAE/g) and irradiated samples (from 7.93 ± 0.21 to 8.26 ± 0.71 mgAAE/g) were similar. Similarly, gamma radiation did not affect the ferric reducing antioxidant power (FRAP). The FRAP values of non-irradiated (89.84 ± 0.59 μmolFeSO4/g) and irradiated samples (from 84.18 ± 1.05 to 88.34 ± 3.35 μmolFeSO4/g) were within the same range.
Total phenolic content
The effect of gamma radiation on total phenolic content in all licorice samples (non-irradiated and irradiated) are shown in Table 2. The total phenolic content of the irradiated samples were within the same range as the control 9.96 ± 1.33 to 11.55 ± 0.84 mgGAE/g. Gamma irradiation at all tested doses (5, 10, 15, and 20 kGy) did not significantly affect changes in the phenolic content in comparison with non-irradiated samples.
Glycyrrhizic acid content
The amount of glycyrrhizic acid, the major phenolic compound in licorice powder, was detected by HPLC (Table 2). The content of glycyrrhizic acid in the non-irradiated control sample was 4.25 ± 0.06, and in irradiated samples, it ranged from 4.15 ± 0.42 to 5.17 ± 0.82 mg/g sample. Significant differences in glycyrrhizic acid content were observed between the irradiated and non-irradiated samples. Samples treated with 15 and 20 kGy showed a significant increase in glycyrrhizic acid, 7.8 and 21.7 percent, respectively.
Table 2. Analysis of antioxidant activities by DPPH and FRAP assays, total phenolic content and glycyrrhizic acid content of licorice extracts at varying irradiation doses.
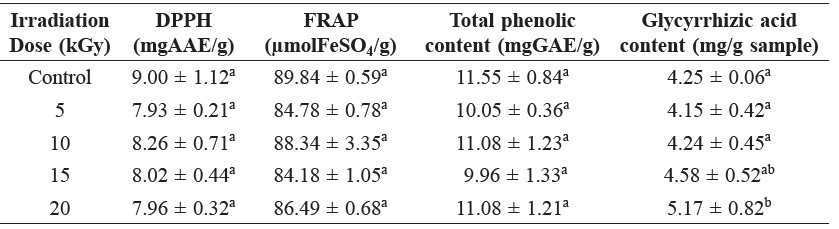
Note: Values are expressed as mean ± SD of triplicate measurements. Different letters in the same column indicate significant differences at p<0.05; AAE = ascorbic acid equivalent, GAE = gallic acid equivalent.
DISCUSSION
Gamma irradiation, a preferred method for reducing microbial contamination, has been approved by the FAO, IAEA, and WHO (WHO, 1981). This work contributed to the knowledge of gamma radiation effects as applied to decontaminating licorice powder. This investigation is among few reports available on the gamma radiation effects on licorice powder in particular, it was the first to report on free radical scavenging activity and glycyrrhizic acid content. We demonstrated the success of decontaminating licorice by gamma irradiation at a dose of 15 kGy without significant loss in licorice’s biochemical properties. In this study, gamma irradiation reduced the total bacterial, yeast, and mold counts linearly with the absorbed doses. These results were in good agreement with those reported by Thomas et al. (2008), who found that irradiation of black tea samples at 7 kGy by gamma radiation led to the desired microbial reduction, while irradiation at 15 kGy resulted in a complete absence of microorganisms. The microorganisms were rendered non-viable by gamma radiation due to various reactive oxygen species (ROS) being produced. ROS introduces lesions in DNA molecules, which are one of the principal targets for radiation damage, preventing the DNA from functioning normally and altering the protein expression (Aouidi et al., 2011).
Gamma radiation at different levels resulted in slight, but non-significant differences in antioxidant activity. Our finding was in agreement with that of Aouidi et al. (2011), who reported the absence of a significant effect of gamma irradiation at 20 kGy on antioxidant activity of air-dried olive leaves. Lee et al. (2005) revealed that gamma irradiation up to 25 kGy did not have any effect on the antioxidant activity of green tea byproducts and green tea leaf extracts. Likewise, Kim et al. (2009) reported that gamma irradiation at 1, 3, 5, and 10 kGy of ethanol extracts of cumin resulted in a slight, but insignificant increase in the antioxidant activity compared with the non-irradiated sample. Our present study accorded well with Koseki et al. (2002), who did not observe any significant changes in the total phenolic content of sweet basil (Ocimum basilicum) at doses up to 30 kGy. This result was similar to Harrison and Were (2007), who showed that irradiated plants exhibited no significant loss of phenolic compounds. Elshazali et al. (2011) obtained a similar result; radiation did not affect tannin content. When the glycyrrhyzic acid content, the major active ingredient of licorice, was analyzed, gamma irradiation led to a significant increase in glycyrrhyzic acid content of the extract. This result correlated with Harrison and Were, 2007 who reported an increase in almond skin phenolic content at 20 and 25 kGy gamma irradiation.
It has been shown that gamma irradiation induces cellulose degradation. Increases in degraded cellulosic products were detected with increasing dose of gamma radiation, when purified cotton linter and purified wood pulp were irradiated (Horio et al., 1963). As cellulose is the principal component of plant cell walls, a high gamma radiation dose should lead to higher extractability of the cellular contents of the plant. Gamma irradiation makes the cell wall more permeable and open, which results in increased diffusion of extracting solvent and enhanced swelling of the cells. This enhanced swelling of the cell wall favors the efficient leaching and extraction of the components (Hussain et al., 2011). As a result, the increased glycyrrhyzic acid content in the extracts we observed could be due to the increased extractability of irradiated licorice powder.
In conclusion, our findings led us to conclude that gamma radiation introduced an important dose-dependent reduction of the bacterial, yeast and molds species in licorice powder, ensuring good microbiological quality, with no significant reduction of antioxidant activity. In addition, gamma radiation at higher doses (15-20 kGy) appeared to enhance the extractability of glycyrrhyzic acid.
REFERENCES
Al-Bachir, M., and G. Lahham. 2003. The effect of gamma irradiation on the microbial load, mineral concentration and sensory characteristics of licorice (Glycyrrhiza glabra L.). Journal of the Science of Food and Agricultural. 83: 70-75. doi: 10.1002/jsfa.1276
Al-Bachir, M., and R. Zeinou. 2005. The effect of gamma irradiation and grinding on the microbial lode of dried licorice roots (Glycyrrhiza glabra L.) and quality characteristics of their extract. Acta Aliment. 34: 287-294. doi: 10.1556/AAlim.34.2005.3.11
Aouidi, F., S. Ayari., H. Ferhi., S. Roussos, and M. Hamdi. 2011. Gamma irradiation of air-dried olive leaves: Effective decontamination and impact on the antioxidative properties and on phenolic compounds. Food Chemistry. 127: 1105-1113. doi: 10.1016/j.foodchem.2011.01.109
Association of Official Analytical Chemists. 1990. Official Method of Analysis (15th ed.). Washington DC.
Benzie, L.F.F., and J.J. Strain. 1996. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Analytical Biochemistry. 239: 70-76. doi: 10.1006/abio.1996.0292
Charpe, T.W., and V.K. Rathod. 2015. Separation of glycyrrhizic acid from licorice root extract using macroporous resin. Food and Bioproducts Processing. 93: 51-57. doi: 10.1016/j.fbp.2013.11.002
Elshazali, A. M., A. A. Nahid, H. A. Salma, and E. B. Elfadil. 2011. Effect of radiation process on antinutrients, protein digestibility and sensory quality of pearl millet flour during processing and storage. International Food Research Journal. 18(4): 1401-1407.
Fargus, J. 1998. Irradiation as a method for decontaminating food: a review. International Journal of Food Microbiology. 44: 189-204. doi :10.1016/S0168-1605(98)00132-9
Harrison, K., and L. M. Were. 2007. Effect of gamma irradiation on total phenolic content yield and antioxidant capacity of Almond skin extracts. Food Chemistry. 102: 932-937. doi: 10.1016/j.foodchem.2006.06.034
Horio, M., I. Rikizo, and M, Hideya. 1963. Effect of Gamma Irradiation upon Cellulose. Bulletin of the Institute for Chemical Research, Kyoto University, 41(1): 17-38.
Hussain, P. R., R.S. Meena, M. A. Dar, and A. M. Wani. 2011. Gamma irradiation of sun dried apricots (Pranus armeniaca L.) for quality maintenance and quarantine purposes. Radiation Physics and Chemistry. 80: 817-827. doi: 10.1016/j.radphyschem.2011.02.029
Khattak, K.F., T.J. Simpson, and Ihasnullah. 2008. Effect of gamma irradiation on the extraction yield, total phenolic content and free radical-scavenging activity of Nigella staiva seed. Food Chemistry. 110: 967-972. doi: 10.1016/j.foodchem.2008.03.003
Khattak, K.F., and T.J. Simpson. 2010. Effect of gamma irradiation on the antimicrobial and free radical scavenging activities of Glycyrrhiza glabra root. Radiation Physics and Chemistry. 79: 507-512. doi: 10.1016/j.radphyschem.2009.10.005
Kim, J. H., M. H. Shin, Y. J. Hwang, P. Srinivasan, J. K. Kim, H. J. Park, et al. 2009. Role of gamma irradiation on the natural antioxidants in cumin seeds. Radiation Physics and Chemistry. 78: 153-157. doi: 10.1016/j.radphyschem.2008.08.008
Kitakawa, I. 2002. Licorice root. A natural sweetener and an important ingredient in Chinese medicines. Pure Applied and Chemistry. 74:1189-1198.
Koseki, P.M., A.L. Villavicencio, M.S. Brito, L.C. Nahme, K.I. Sebastiao, , P.L. Reta, et al. 2002. Effects of irradiation in medicinal and eatable herbs. Radiation Physics and Chemistry. 63: 681-684.
Lee, J., R.W. Durst, and R.E. Wrolstad, 2005. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: collaborative study. Journal of AOAC International. 88: 1269-1278.
Mustapha, M.B., M. Bousselmi, T. Jerbi, N.B. Bettaieb, and Fattouch, S. 2014. Gamma radiation effects on microbiological, physico-chemical and antioxidant properties of Tunisian millet (Pennisetum glaucum L.R.Br.). Food Chemistry. 154: 230-237. doi: 10.1016/j.foodchem.2014.01.015
Shin, Y.W., E.A., Bae, B. Lee, S.H. Lee, J.A. Kim, Y.S. Kim, and D.H. Kim, 2007. In vitro and in vivo antiallergic effects of Glycyrrhiza glabra and its componenets. Planta Medicine. 73: 257-261. doi: 10.1055/s-2007-967126
Thomas, J., R.S. Senthikumar, R.R Kumar, A.K. Mandal, and N. Muraleedharan. 2008. Induction of γ–irradiation for decontamination and to increase the storage stability of black teas. Food Chemistry. 106: 180-184. doi: 10.1016/j.foodchem.2007.05.064
Vaya, J., P.A. Belinky, and M. Aviram. 1997. Antioxidant constitute from licorice roots: isolation, structure elucidation and antioxidant capacity toward LDL oxidation. Free Radical Biology and Medicine. 23: 302-313.
Velioglu, Y.S., G. Mazza, L. Gao, and B.D. Oomah. 1998. Antioxidant capacity and total phenolics in selected fruits, vegetable and grain products. Journal of Agricultural and Food Chemistry. 46: 4113-4117. doi: 10.1021/jf9801973
WHO. 1981. Wholesomeness of irradiated food: report of the Joint FAO/IAEA/WHO Expert Committee. In :World Health Organization Technical Report Series 659. WHO, Geneva, Switzerland. doi: 10.1016/0147-6513(80)90017-2
World Health Organization. 1998. Life in the 21st century a vision for all. Report of the Director-General.
Wachiraporn Pewlong*, Surasak Sajjabut, Jaruratana Eamsiri, Sirilak Chookaew and Kanokporn Boonsirichai
Nuclear Research and Development, Thailand Institute of Nuclear Technology, Ongkharak, Nakhon Nayok 26120, Thailand
*Corresponding author. E-mail: wachiraporn03@yahoo.com
Total Article Views

