
Importance of Tien Yen Estuary (Northern Vietnam) for Early-Stage Nuchequula nuchalis (Temminck & Schlegel, 1845)
Hau Duc Tran*, Thuy Thi Ta and Thanh Trung TranPublished Date : 2019-08-23
DOI : 10.12982/cmujns.2016.0006
Journal Issues : Number1 ,January - April 2016
ABSTRACT
To understand the importance of an estuary for the early stage of Nuchequula nuchalis (Spotnape Ponyfish), we collected samples (5,430 individuals) from the shallow areas of the Tien Yen estuary, northern Vietnam, from March 2013 to February 2014. The water temperature, salinity and turbidity at the collection sites varied from 18 to 29°C, 0 to 19 psu, and 4 to 96 NTU, respectively. The fish caught were mainly postflexion larvae, with a few juveniles. While the fish occurred from April 2013 to January 2014, they were heavily concentrated during two months only – April and May 2013, when more than 99% of the postflexion larvae were collected. The fish were distributed mostly in the middle part of the estuary, implying that this area is its main habitat during the early stages of its lifecycle.
Keywords: Nuchequula nuchalis, Larvae and juveniles, Occurrence, Estuarine habitat, Northern Vietnam
INTRODUCTION
Nuchequula nuchalis (Spotnape Ponyfish) is distributed in East Asia (Kimura et al., 2008b), including Taiwan and from south of Fujian to the Gulf of Tonkin (Woodland et al., 2001); this probably includes northern Vietnamese coastal waters. This species, with others in the Leiognathidae family, are commercially important in Asian wild fisheries and aquaculture, and inhabit coastal and estuarine waters (Woodland et al., 2001). Occurrence of the early stage of N. nuchalis has only been recorded in northern Kyushu, Japan, and its spawning season is from the middle of May to the end of July (Fujita, 1960); little is known about the distributional pattern of the early stages of this fish in an estuary.
Tran and Ta (2014a) recorded 193 fish species, belonging to 142 genera and 83 families, in the Tien Yen estuary. Some research has been conducted on the early life stages of fish in this estuary, such as descriptions of Sillago sihama (Tran et al., 2014) and Nuchequula nuchalis (Tran et al., 2014). The distribution patterns of some species have also been elucidated, namely Oryzias curvinotus (Ta et al., 2014) and species of Lateolabrax (Tran and Ta, 2014b); it is presumed that the Tien Yen estuary plays a crucial role as their nursery ground. It has also been shown that fish populations in the Tien Yen River have been dramatically reduced recently, partly due to anthropogenic pressures (Tran and Ta, 2014a). Thus, understanding early-stage fish recruitment in this estuary is a priority to prevent further reduction and/or promote replenishment.
Recently, a number of larvae and juveniles belonging to the Nuchequula genus were collected along the shore of the Tien Yen estuary, northern Vietnam; based on morphology, they were identified as (Tran et al., 2014). This study reports occurrence data of the larvae and juveniles of N. nuchalis in this northern Vietnam estuary, probably the southernmost distribution area that has been recorded.
MATERIALS AND METHODS
Spotnape ponyfish larvae and juveniles were sampled monthly from the Tien Yen estuary, northern Vietnam, from March 2013 to February 2014 (Figure 1). The river, 82 km long, flows into the northern Vietnam coast on the Gulf of Tonkin; the headwaters are partly located in China. The maximum width of the estuary is 2 km and its maximum depth is approximately 8 m. The estuary is characterized by a large tidal flat, with average tidal fluxions of 3-4 m (Vu, 2009). The salt wedge can reach approximately 15 km upriver from the river mouth. Sampling stations (TS2-TS9) were chosen to represent the entire estuary, and the uppermost station was located at the end of the tidal basin (Figure 1). A simultaneous collection (TS1) was made at the outer estuary as a control site.
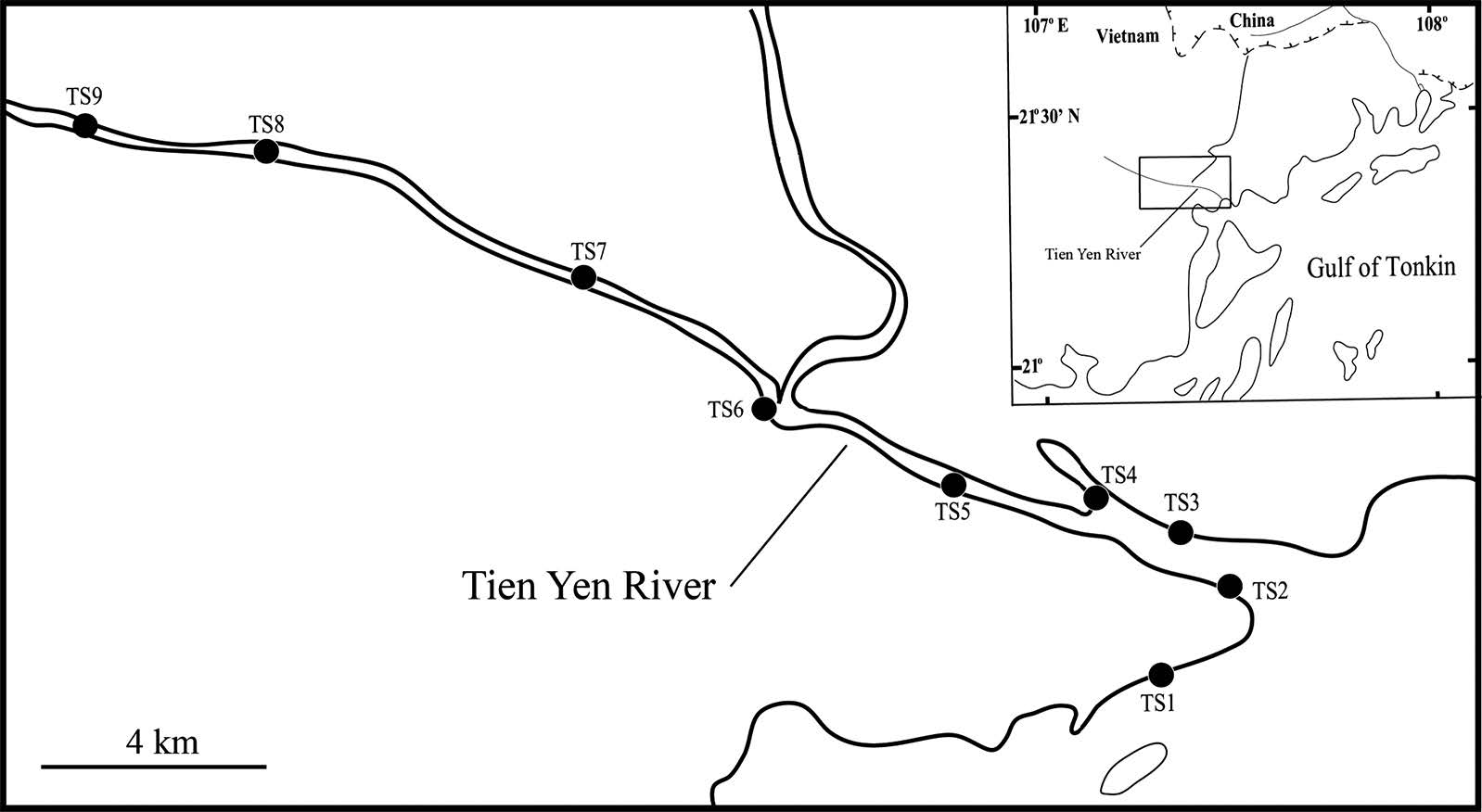
Figure 1. Location and sampling stations in the Tien Yen estuary. Stations where Spotnape Ponyfish were sampled are indicated by solid circles (stations TS1–TS9).
Collections were made in shallow areas of the Tien Yen estuary by a small seine net (1 × 4 m, 1 mm mesh-aperture) (Kinoshita et al., 1988). Two persons kept the net stretched, and waded backward in the waters, from ankle- to neckdepth along the shoreline for a distance of ca. 50 m (2 min.). A day’s collection usually consisted of one to four hauls at each bank water station. Catch Per Unit of Effort (CPUE) is the number of individuals in each haul (ca. 2 minutes or 50 m). All specimens were fixed in 10% formalin, and sorted specimens were transferred to 80% ethanol. Water temperatures (°C), salinities (psu) and turbidity (NTU) were measured at each station during the sampling periods using a Water Quality Checker (WQC-22A, TOA DKK – temperature: 0-50°C ± 0.1°C; salinity: 0-4psu ± 2.5%; turbidity: 0-800NTU ± 0.1%).
The N. nuchalis specimens were divided into developmental stages, in accordance with Kendall et al. (1984). Standard length was measured to the nearest 0.1 mm using an ocular micrometer attached to a stereo microscope. In this study, standard length indicates postflexion larvae and juveniles.
The linear relationships between paired quantitative variables, namely CPUE and temperature, CPUE and salinity, or CPUE and turbidity, were ascertained using the Spearman’s Rho rank correlation, because these data, tested by Shapiro-Wilk (n < 50), were non-normal curve distributions. The value of Spearman rank (r) is from -1 to + 1. A value closer to 1 indicates strong agreement, while r closer to -1 indicates strong agreement in the reverse direction. In Figures 2 and 3, the CPUE was lg (x+1)-transformed.
RESULTS
Seasonal changes of N. nuchalis along the shallow waters of the Tien Yen estuary from March 2013 to February 2014 are described in Table 1. Nearly all (99.5%) of the specimens were collected in April and May. No fish were collected in March, then the average CPUE increased remarkably to 115.8 individuals per haul in April, reaching a peak in May of 459.3 individuals per haul. Thereafter, the value fluctuation from June to February varied from 0.1 to 2.1 individuals per haul (Table 1); with no specimens collected in June, November, and February. The average fish size increased from April to July, then decreased in August, before remaining roughly constant until January (Table 1). The size range varied the most in May. The fish collected in April were the smallest, with a body length of around 7.0 mm SL (Table 1).
Average water temperature was 23.8°C in March 2013, then rose to 29.4°C in September, before declining to 17.3°C in January 2014; eventually, increasing to 20.6°C in February (Table 1). The water temperature was higher than 24°C from March to November 2013, and lower than 21°C from December 2013 to February 2014 (Table 1). On the other hand, the average salinity fell from around 16.8 psu in March, to approximately 3 psu in May to August, after which, the salinity increased to 11.5 psu by November, and subsequently stabilized around 12.7 psu until February (Table 1). The monthly relationship between the temperature and salinity showed a reciprocal pattern (r = -0.75, p < 0.05) (Table 1). The turbidity showed a different pattern, with extremely turbid water (150 NTU) in May (Table 1). In some other months, turbidity was less than 20 NTU (Table 1). The monthly relationship between salinity and turbidity also showed a negative pattern, but the difference was not significant (r = -0.68, p > 0.05) (Table 1). Additionally, tidal changes did not show a connection with the fish collection during the study period (Table 1).
Table 1. Environmental parameters, CPUE and body length of N. nuchalis larvae and juveniles collected in Tien Yen estuary during March 2013 to February 2014.
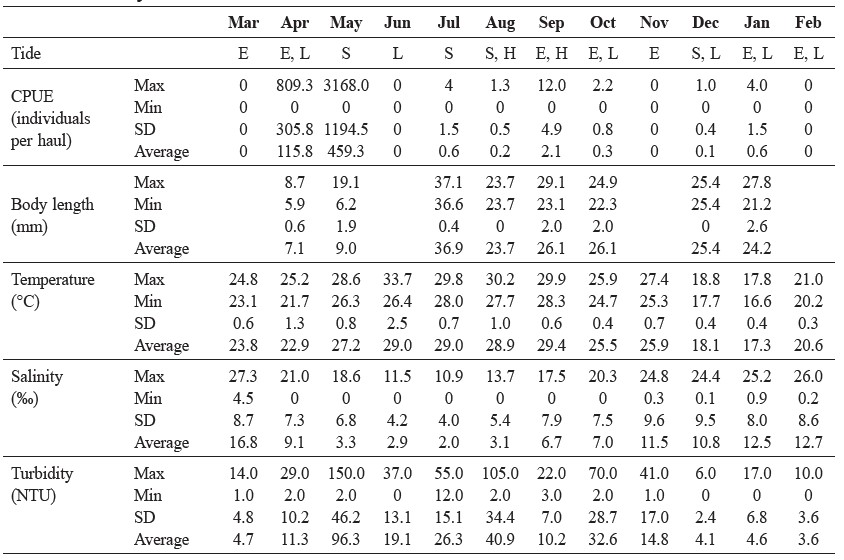
Note: E. ebb tide, L. low tide, S. spring tide, H. high tide.
Because none of the larvae or juveniles were caught in stations TS8 and TS9, where the salinity was 0 psu during the study period, this paper omits the above two stations in the next parts (Figure 2). Among the seven stations from TS1 to TS7, fish occurred abundantly at stations TS4 and TS5 – the middle part of the estuary – the average CPUE was 264.4 and 77.8 individuals per haul, respectively. In contrast, the average CPUE ranged from 0.06 to 0.9 individuals per haul at stations TS1, TS2, TS3, TS6, and TS7 (Figure 2). The average fish size reduced slightly from TS1 to TS5 (Figure 2). Only one specimen was collected at station TS6 (37.1 mm SL). The size of specimens collected at stations TS4 and TS5 was larger than those collected at the other stations (Figure 2).
A few differences in the average water temperature were found among the seven stations (TS1–TS7), while the average salinity showed a significantly different pattern from the temperature (Figure 2). Salinity was highest in the outer stations (TS1, TS2), where the tidal effects were largest, and decreased moving upstream; the salinity was less than 10 psu from stations TS3 to TS7, with TS5-TS7 at ca. 1 to 5 psu. The average turbidity fluctuated among the seven stations; it was lowest at station TS1 (16 NTU) and highest at station TS5 (29.5 NTU) (Figure 2).
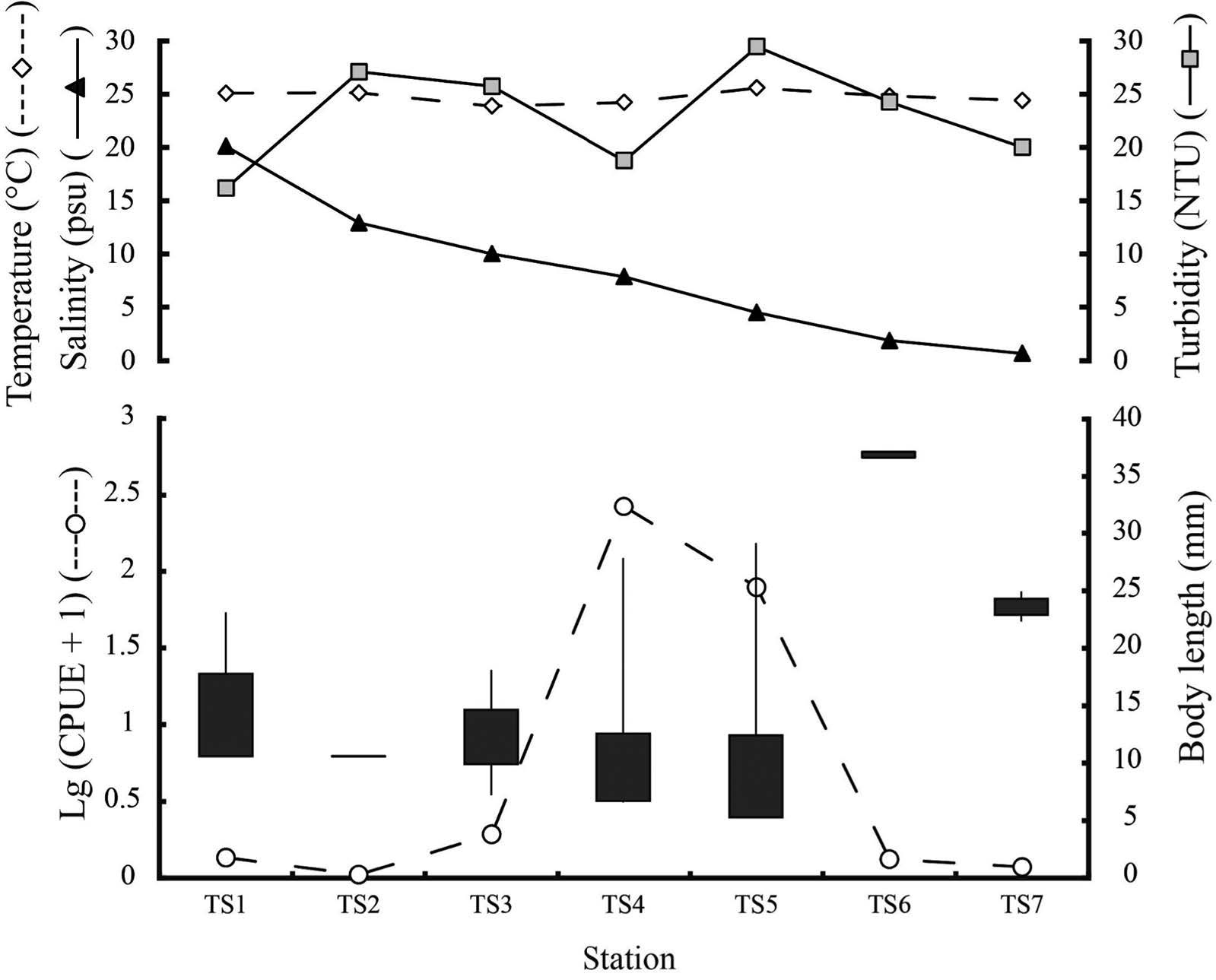
Figure 2. Spatial variation of N. nuchalis average CPUE collected in the bank waters of the Tien Yen estuary, during March 2013 to February 2014. Average water temperature, salinity, and turbidity are shown by diamond, triangle, and square, respectively. Vertical lines and thick bars denote ranges of and average ±SE of standard length, respectively.
DISCUSSION
Nuchequula nuchalis occurred in the Tien Yen estuary during two periods – from April to July 2013 and from August 2013 to January 2014, with a peak in May 2013. The spawning season of this fish in northern Vietnam may take place earlier, and last longer than in northern Kyushu, Japan, where the breeding season is from middle May to the end of July (Fujita, 1960). In Japan, Equulites rivulatus larvae appear in the summer, showing a greater deviation in spawning months at lower latitudes (Haque and Ozawa, 1995b), unlike that in sparid fish. At higher latitudes, various species of sparids spawn year round, in contrast to lower latitudes, where spawning correlates with the months of lowest sea surface temperature (Sheaves, 2006). The spawning period of N. nuchalis reported in this study showed a similar tendency as that of Equulites rivulatus. Smaller collections in April and May were from around 5.0 mm SL (Table 1), and in the laboratory, hatching larvae were 1.4 mm in total length (Fujita, 1960). Based on this, the peak of the spawning season of the fish in this study was estimated in March or April, during which the average water temperatures at the river bank were lower than 25°C (Table 1). As can be seen from Table 1 and Figure 2, the average water temperature and CPUE did not show a positive relationship by month (r = 0.07) or by station (r = -0.28). Therefore, the assemblages of this fish larvae and juveniles in the bank waters of the estuary seem to be unrelated to changes in water temperature.
The peak of occurrence of the N. nuchalis larvae and juveniles was in May, when average turbidity was highest (Table 1). However, although the average turbidity and fish caught were positively related by month, the difference was not significant (r = 0.41, p > 0.05) (Table 1), and there was no relationship by stations (r = 0) (Figure 2). There is no evidence that turbidity influenced the distribution of Leiognathus equulus fish in northeastern Queensland, Australia (Johnston et al., 2007), although turbidity provided early stages with cover from predators and could be beneficial for increasing benthic production of fish (Cyrus and Blaber, 1987). While May, the month with the highest turbidity, accounted for more than 99% of the fish collected over the study period, station TS4, at which the turbidity was only 2 NTU (the lowest of the seven stations), yielded 99% of May’s total specimens (Figure 3). Hence, factors other than water temperature and turbidity may contribute to the distribution of this fish along the bank waters of the estuary.
While the occurrence of N. nuchalis larvae and juveniles might be related to the stations along the estuary, the Spearman’s correlation coefficient between salinity and CPUE at the seven stations is -0.14 (Figures 2, 3). Most of specimens were collected in the middle part of the estuary (stations TS4 and TS5), at which the average salinity ranged from 3 to 8 psu (Figure 2). At station TS4, the peak catch occurred in May, when the salinity reached only 0.3 psu, while another peak appeared in April at station TS5, when the salinity was just 2.1 psu (Figure 3). At stations TS6 and TS7, only larger specimens were collected; this implies that larger juveniles were more tolerant of freshwater (Figures 2, 3). In contrast, at station TS1 at the mouth of the estuary, where average salinity was around 20 psu, few larvae and juveniles were collected (Figures 2 and 3). This finding indicates that the variation in salinity along the Tien Yen estuary could be one of the factors that influence the occurrence of N. nuchalis larvae and juveniles. This phenomenon was also observed with Liza richardsonii, L. dumerili, and Mugil cephalus (Whitfield, 1994). However, despite some other months having lower salinity than April and May, few specimens were collected (Figure 3). Therefore, occurrence of the early stage of N. nuchalis in the estuary was partly driven by its reproductive cycle, concentrated in March or April (Table 1).
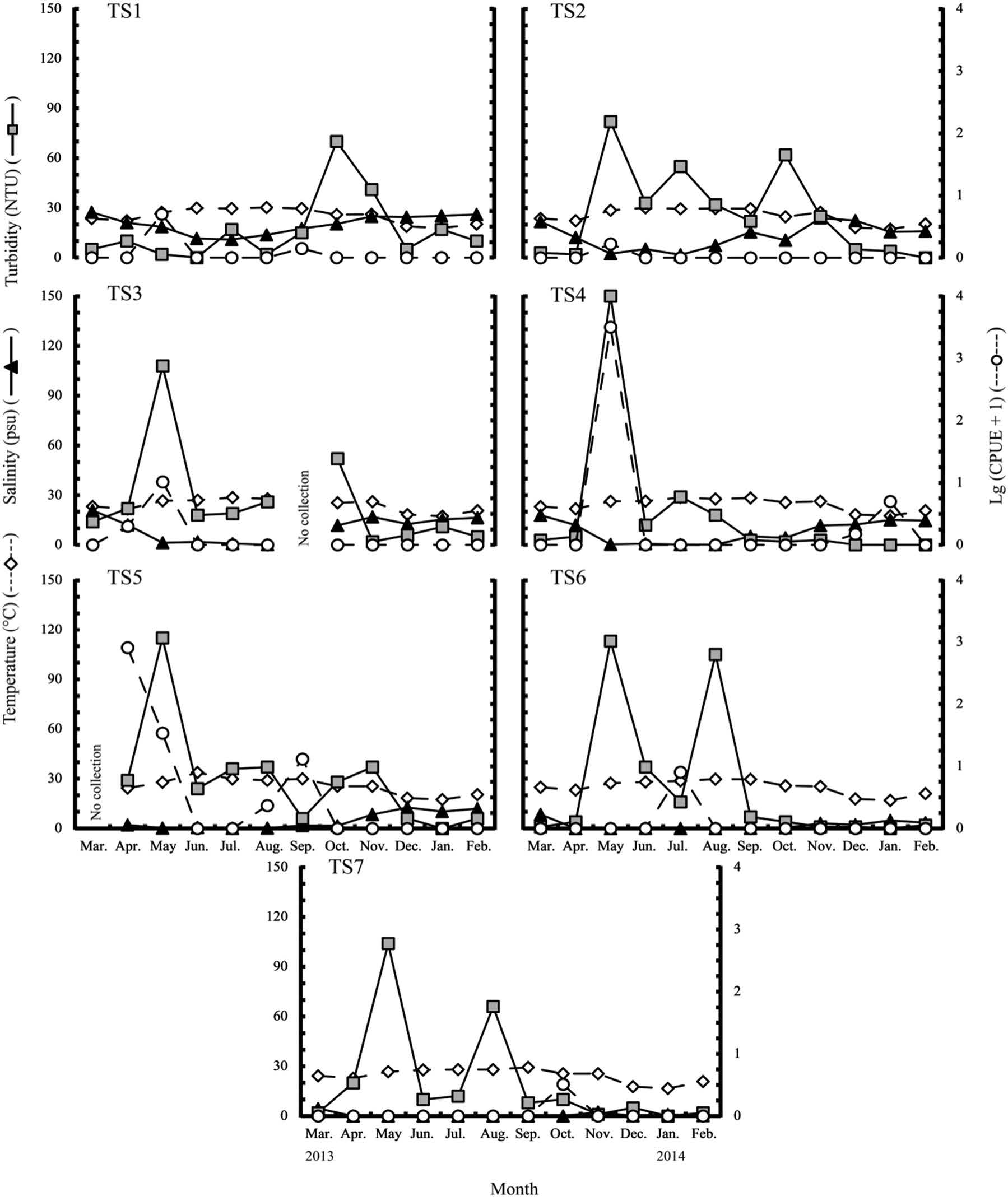
Figure 3. Comparison of the CPUE of Spotnape Ponyfish and water conditions among seven stations in the Tien Yen estuary, during March 2013 to February 2014. Otherwise, same as in Figure 2.
This study confirmed that the Tien Yen River plays a crucial role for in the early stages of N. nuchalis’s lifecycle (Ta et al., 2014; Tran and Ta, 2014b). Recently, overfishing, destructive fishing methods, water pollution, and poor management have placed considerable pressure on the Tien Yen estuary fish resources (Tran and Ta, 2014a), possibly affecting the early life stages as well. Hence, more attention needs to be paid to preserving the estuary and its fishery resources.
This study yielded a number of larval and juvenile along the bank waters of the Tien Yen estuary in northern Vietnam during March 2013 to February 2014, with the vast majority found in May in the middle part of the estuary. Salinity is a possible factor influencing the occurrence of the larval and juvenile stages of N. nuchalis in an estuarine environment.
ACKNOWLEDGMENTS
The authors are grateful to Ken Maeda for his critical reading of the initial manuscript and two anonymous reviewers for their helpful comments. The Nagao Natural Environment Foundation (Japan), financially supported this study, through the second author. All the surveys in the present study comply with the current laws of Vietnam.
REFERENCES
Cyrus, D.P., and S.J.M. Blaber. 1987. The influence of turbidity on juvenile fishes in estuaries, Part 1, Field studies at Lake St Lucia on the southeastern coast of Africa. Journal of Experimental Marine Biology and Ecology 109: 53-70.
Fujita, S. 1960. Egg development and prelarval stages of a silver belly, Leiognathus nuchalis (Temminck et Schlegel). Bulletin of the Japanese Society for Science of Fish 26: 1091-1094.
Haque, M.M., and T. Ozawa. 1995a. Ontogenetic larval characters of three Leiognathid species in Kagoshima Bay, Southern Japan. Japanese Journal of Ichthyology 42: 137-146.
Haque, M.M., and T. Ozawa. 1995b. Occurrence and abundance of Leiognathus rivulatus (Temminck and Schlegel) larvae in Kagoshima Bay, Southern Japan, in 1991. Japanese Journal of Ichthyology 42: 131-136.
Johnston, R., M. Sheaves, and B. Molony. 2007. Are distributions of fishes in tropical estuaries influenced by turbidity over small spatial scales? Journal of Fish Biology 71(3): 657-671.
Jones, G. 1985. Revision of the Australian species of the fish family Leiognathidae. Australian Journal of Marine & Freshwater Research 36: 559-613.
Kendall, A.W., E.H.Jr. Ahlstrom, and H.G. Moser. 1984. Early life history stages of fishes and their characters. In Moser H.G., Richard W.J., Cohen D.M., Fahay M.P., Kendall A.W.Jr. and Richardson S.L., eds., Ontogeny and Systematics of Fishes. American Society of Ichthyologists and Herpetologists, Special Publication 1: 11-22.
Kimura, S., K. Ikejima, and Y. Iwatsuki. 2008a. Eubleekeria Fowler 1904, a valid genus of Leiognathidae (Perciformes). Ichthyological Research 55: 202-203.
Kimura, S., R. Kimura, and K. Ikejima. 2008b. Revision of the genus Nuchequula with descriptions of three species (Perciformes: Leiognathidae). Ichthyological
Research 55: 22-42.
Kinoshita, I. 1988. Leiognathidae. in: Okiyama M., ed., An atlas of the early stage fishes in Japan, Tokai University Press, Tokyo, Japan: 483-486.
Kinoshita, I., S. Fujita, I. Takahashi, and Azuma K. 1988. Occurrence of larval and juvenile Japanese snook, Lates japonicus, in the Shimanto estuary, Japanese Journal of Ichthyology 34: 462-467.
Myoung, J.G. and Y.U. Kim. 1984. Morphology of larvae and juveniles of Leiognathus nuchalis (Temminck et Schlegel). Bulletin of the National Fisheries University of Pusan 24: 1-22.
Nguyen, V.H. 2005. Freshwater fish of Viet Nam. Agricultural Publishing House, Hanoi.
Sheaves, M. 2006. Is the timing of spawning in sparid fishes a response to sea temperature regimes? Coral Reefs 25: 655-669.
Sparks, J.S., P.V. Dunlap, and W.L. Smith. 2005. Evolution and diversification of a sexually dimorphic luminescent system in ponyfishes (Teleostei: Leiognathidae), including diagnoses for two new genera. Cladistics 21: 305-327.
Ta, T.T., T.T. Hoang, T.T. Tran, and D.H. Tran. 2014. Distribution of Oryzias curvinotus in Tien Yen estuary, Quang Ninh province, Vietnam. Journal of Science, Vietnam National University, Hanoi, Natural Science and Technology 30: 235-241.
Tran, D.H., and T.T. Ta. 2014a. Fish diversity and fishery status in the Ba Che and Tien Yen Rivers, northern Vietnam, with consideration on factors causing recent decline of fishery products. Kuroshio Science 7: 113-122.
Tran, D.H., and T.T. Ta. 2014b. Distribution of larvae and juveniles of Lateolabrax sp. in Tien Yen and Kalong Rivers, Vietnam. Journal of Science, Vietnam National University, Hanoi, Natural Science and Technology 30 (6S): 137-142.
Tran, D.H., T.T. Nguyen, and T.T. Ta. 2014. Morphological description of larvae and juveniles of Sillago sihama (Forsskal) from the Tien Yen estuary, Vietnam. Journal of Science, Vietnam National University, Hanoi, Natural Science and Technology 30: 58-63.
Tran, T.T, D.H. Tran, and T.T. Ta. 2014. The larval and juvenile morphology of Nuchequula nuchalis (Temminck & Schlegel, 1845). Journal of Science of HNUE 59: 117-124.
Vu, T.T. 2009. Estuarine ecosystems in Vietnam. Educational Publishing House, Hanoi.
Whitfield, A.K. 1994. Abundance of larval and 0+ juvenile marine fishes in the lower reaches of three southern African estuaries with differing freshwater inputs. Marine Ecology Progress Series 105: 257-267.
Woodland, D.J., S. Premcharoen, and A.S. Cabanban. 2001. Leiognathidae. Slipmouths (ponyfishes). p. 2791-3380. In Carpenter K.E. and Niem V.H., eds. FAO species identification guide for fishery purposes. The living marine resources of the Western Central Pacific, Volume 5, Bony fishes part 3 (Menidae to Pomacentridae), Rome, FAO.
Hau Duc Tran1*, Thuy Thi Ta2 and Thanh Trung Tran1
1 Hanoi National University of Education, 136 Xuan Thuy, Cau Giay, Hanoi, Vietnam
2 Hanoi Metropolitan University, 98 Duong Quang Ham, Cau Giay, Hanoi, Vietnam
*Corresponding author. E-mail: hautd@hnue.edu.vn
Total Article Views

