
35% Carbamide Peroxide: A Threat to Male Fertility in Wistar Rats
Olugbemi T. Olaniyan*, Olorunsola I. Adeyomoye, Gloria E. Okotie, Femi Adebayo, and Ibitoye O BabatundePublished Date : March 12, 2025
DOI : https://doi.org/10.12982/NLSC.2025.036
Journal Issues : Number 2, April-June 2025
Abstract This study examined the impact of 35% carbamide peroxide on male reproductive functions in Wistar rats. Carbamide peroxide, commonly found in tooth-whitening products, has known oral effects, but its reproductive implications are not fully understood. Twenty-four Wistar rats were divided into four groups (n=6): a control group and three test groups receiving carbamide peroxide at 100 mg/kg, 250 mg/kg, and 500 mg/kg respectively and orally for 21 days. After administeration, the rats were sacrificed, and blood samples were analyzed for follicle-stimulating hormone (FSH), luteinizing hormone (LH), and testosterone via ELISA. Body and testicular weights, malondialdehyde (MDA) levels, sperm motility, count, and morphology were measured using standard procedures, while testicular histology was assessed through hematoxylin and eosin staining. Data were expressed as mean ± SEM at P<0.05. Results indicated a significant decrease in sperm motility and FSH levels in 100 mg/kg and 250 mg/kg carbamide peroxide groups, while MDA levels increased significantly in the 250 mg/kg and 500 mg/kg carbamide peroxide groups when compared to control. Histological analysis showed testicular hypertrophy and epididymal fat deposition in carbamide peroxide groups at 100 mg/kg and 500 mg/kg. Oral carbamide peroxide exposure led to reduced FSH levels and sperm motility, along with increased MDA concentration, fat deposition and testicular enlargement, suggesting detrimental effects on male reproductive health.
Keywords: Carbamide peroxide, Sperm motility, Follicle stimulating hormone, Malondialdehyde, Male reproductive functions
Citation: Olaniyan, O.T., Adeyomoye, O.I., Okotie, G.E., Adebayo, F., and Babatunde, I.O. 2025. 35% Carbamide peroxide: A threat to male fertility in wistar rats. Natural and Life Sciences Communications. 24(2): e2025036.
INTRODUCTION
The male reproductive system plays a vital role in maintaining its functions through the coordinated action of various structures, including the testes, epididymis, vas deferens, prostate, bulbourethral glands, scrotum, and penis. These organs are essential for spermatogenesis, androgen synthesis, and the development of male sexual characteristics, as well as sexual function (Cannarella et al., 2020; Haidl, 2024). Testosterone, the primary male sex hormone, is produced by Leydig cells located in the interstitial space of the testes. This hormone can be further metabolized into dihydrotestosterone (DHT) by 5-alpha reductase in the prostate or into estradiol via aromatase, an enzyme found in adipose tissue (Huijben et al., 2023). Testosterone, DHT, and estradiol are steroid hormones that exert their biological effects by diffusing through the cell membrane and regulating intracellular protein expression (Ide, 2023).
Testosterone production is regulated by the hypothalamic-pituitary-gonadal (HPG) axis. Gonadotropin-releasing hormone (GnRH) from the hypothalamus stimulates the anterior pituitary to release luteinizing hormone (LH) and follicle-stimulating hormone (FSH), which act on the testes to promote spermatogenesis and androgen production (Khumsap et al., 2024). Disruptions in this axis, particularly through hormonal imbalances, can impair reproductive function. For example, excessive conversion of testosterone to estradiol has been linked to pathological conditions such as osteoporosis, gynecomastia, and infertility (Daniel et al., 2015).
Male infertility is a significant concern worldwide and is defined as the inability of a couple to conceive after one year of regular, unprotected intercourse (Odetayo et al., 2024). Several factors contribute to male infertility, including hormonal imbalances, testicular defects, abnormal sperm morphology, and environmental exposures (Deshpande and Gupta, 2019). Despite the common perception that infertility is primarily a female issue, male factors contribute to nearly half of all infertility cases (Öztekin et al., 2019). A thorough evaluation of the male partner is therefore essential for effective diagnosis and treatment, which may involve the use of assisted reproductive technologies (ARTs) (GBD, 2024).
In recent years, there has been a growing demand for tooth-whitening products due to increased awareness and aesthetic desires (Jin et al., 2024). Many of these products contain carbamide peroxide, which decomposes into hydrogen peroxide, a potent bleaching agent (Peixoto et al., 2018). However, the widespread use of carbamide peroxide has raised concerns regarding its potential side effects. Studies have reported that peroxide-based whitening agents can increase tooth sensitivity, cause gingival irritation, and lead to enamel demineralization and pulp damage (Albanai et al., 2015). More alarmingly, there has been reported genotoxic, carcinogenic, and gastric mucosal toxicities associated with these products (Asgari et al., 2024). Despite these risks, their global demand continues to rise. Considering the potential for carbamide peroxide to induce oxidative stress, which has been implicated in reproductive dysfunctions, this study aims to investigate the effects of 35% carbamide peroxide, commonly found in tooth-whitening products, on male reproductive function using Wistar rats as an experimental model.
MATERIALS AND METHODS
Materials
The materials used for this study included rat cages, sawdust (for bedding), feeding and drinking cans, animal feed, hand gloves, a light microscope (Olympus, Japan), distilled water, pipettes, a weighing scale, microscopic slides, neutral buffered formalin, 35% carbamide peroxide, and hormonal assay kits for follicle-stimulating hormone (FSH), luteinizing hormone (LH), and testosterone. The ELISA kits used for hormonal assays were obtained from Bioassay Technology Laboratory (BT Lab), China, with the following batch/lot numbers: FSH (E0010Hu, Lot: 20240101), LH (E0007Hu, Lot: 20240102), and Testosterone (E0004Hu, Lot: 20240103).
Experimental animals
Twenty-four male Wistar rats, weighing between 100 and 120 g, were obtained from the Animal House of the Faculty of Basic Medical Sciences, University of Ibadan, Oyo State, Nigeria. The rats were housed in the animal facility at Bingham University, Nasarawa State, Nigeria, where they were acclimatized for two weeks before the commencement of the experiment. During this period, the rats were maintained on a standard rat diet obtained from Ladokun Feed Limited, Ibadan, and provided with tap water ad libitum under hygienic conditions.The housing facility was well-ventilated, with the temperature maintained at 27°C, and a 12-hour light/dark cycle was implemented. The animals were housed in plastic cages (6 rats per cage), and the bedding (wood shavings) was changed every two days.
Ethical considerations
This study was approved by the Animal Ethics Committee of Bingham University, Karu, Nasarawa State, Nigeria. All experimental procedures were conducted in accordance with the guidelines outlined by the National Institutes of Health for the care and use of laboratory animals (Baker et al., 2019) and in line with the Declaration of Helsinki.
Animal grouping and study design
The rats were randomly assigned to four groups, each comprising six animals:
Group 1 (Control): Received distilled water.
Groups 2, 3, and 4: Administered with carbamide peroxide at doses of 100 mg/kg, 250 mg/kg, and 500 mg/kg, respectively (Ferreira Neves Dias et al., 2022).
The carbamide peroxide was dissolved in distilled water and administered orally to the test groups once daily for 21 days. The doses of carbamide peroxide at 100 mg/kg, 250 mg/kg, and 500 mg/kg, respectively were selected based on their potential to produce a graded dose-response, allowing for the evaluation of dose-dependent effects on male reproductive function. These doses span a low, moderate, and high range to explore toxicity thresholds and therapeutic margins. A 21-day duration is sufficient to assess the subchronic impacts on reproductive tissues and sperm parameters, as spermatogenesis typically takes around this time in rodents. Testes and Body weights were recorded at the end of the experiment. The relative weight of the testes was calculated by dividing the testes weight by the body weight and then multiplying the result by 100. This study utilized a sample size of n=6 per group (total N=24) due to practical constraints such as resource limitations and ethical considerations for minimizing animal use. While the achieved power (21.6%) is lower than the recommended 80%, this study aims to provide preliminary insights into the effects of carbamide peroxide. The effect size (f=0.25) was chosen based on prior literature, and future studies with larger sample sizes will be conducted to validate these findings. Results should be interpreted with caution, particularly for nonsignificant outcomes.
Animal sacrifice and tissue collection
On day 21, following the administration period, the animals were sacrificed by cervical dislocation. A midline abdominal incision was made, and the testes were harvested. The epididymis were carefully dissected from the testes, blotted dry, and preserved in Bouin's fluid for histological analysis. The testes were weighed after the removal of surrounding tissues.
Sperm count determination
Sperm density was assessed by homogenizing the cauda epididymis in a physiological solution (Sigma-Aldrich, USA) and allowing the spermatozoa to disperse at room temperature for 24 hours (Li et al., 2023). The sperm suspension was diluted (1:11), and 10 µL was loaded into a Mallassez hemocytometer (Marienfeld, Germany). Spermatozoa were counted in 20 randomly selected squares using a light microscope (Olympus, Japan, 40x objective lens), and sperm density was expressed as the total number of spermatozoa.
Sperm motility assessment
Immediately after sacrifice, the right cauda epididymis was minced in 10 mL of saline solution (0.9%) (Sigma-Aldrich, USA) at 34°C. Sperm motility was assessed by placing 10 µL of the diluted solution into the chambers of a Mallassez hemocytometer (Marienfeld, Germany). Motile and non-motile spermatozoa were counted in 10 random squares using a light microscope (Olympus, Japan, 40x objective lens), and motility was expressed as a percentage of motile spermatozoa (Adamkovicova et al., 2016).
Sperm morphology assessment
Sperm morphology was evaluated by preparing a smear from the cauda epididymis solution. The smear was stained with eosin-necrosin (Sigma-Aldrich, USA) and air-dried. A total of 200 spermatozoa were examined in 20 random fields under a light microscope (Olympus, Japan), and the percentages of spermatozoa with morphological abnormalities were recorded. Non-viable spermatozoa, indicated by stained cytoplasm, were also counted, and the percentage of viable spermatozoa was calculated (Adamkovicova et al., 2016).
Blood collection and serum preparation
Blood samples were collected via retro-orbital sinus puncture using capillary tubes and placed into plain tubes. The blood was centrifuged at 700 × g for 5 minutes to separate the serum, which was used for malondialdehyde (MDA) determination and hormonal assays.
Determination of MDA level
Malondialdehyde (MDA) levels were determined using the thiobarbituric acid reactive substances (TBARS) method, as described by Stocks and Domandy (Vukšić et al., 2023). In this procedure, 0.1 mL of serum samples were mixed with 1 mL of 20% trichloroacetic acid (Sigma-Aldrich, USA) to precipitate proteins, followed by centrifugation using a centrifuge (Eppendorf 5430R, Germany) to obtain the supernatant. The supernatant was then reacted with thiobarbituric acid (Sigma-Aldrich, USA), forming a pink MDA-TBA complex. The absorbance of this complex was measured at 532 nm using a spectrophotometer (Shimadzu UV-1800, Japan).
Hormonal assay
For the determination of FSH (Follicle-Stimulating Hormone), 100 µL of serum was used as the sample volume, with incubation at 37°C to optimize binding between FSH and specific antibodies on the ELISA plate (Bioassay Technology Laboratory, China, Lot: E0010Hu). After incubation, a substrate solution (Sigma-Aldrich, USA) was added, and the absorbance was measured at 450 nm using a spectrophotometer (Shimadzu UV-1800, Japan). Hormone concentrations were calculated using a standard curve from known concentrations. The procedure for LH (Luteinizing Hormone) followed the same steps, using the ELISA kit (Bioassay Technology Laboratory, China, Lot: E0007Hu) with 100 µL of serum, incubation at 37°C, absorbance reading at 450 nm, and determination of LH levels using a standard curve. For testosterone, a smaller sample volume of 50 µL was used, incubated at 37°C for optimal binding on the ELISA plate (Bioassay Technology Laboratory, China, Lot: E0004Hu). The absorbance was measured at 450 nm using the Shimadzu UV-1800 spectrophotometer, and testosterone concentrations were calculated using a standard curve (Burow et al., 2019).
Histological procedures
Testicular tissues were processed by dehydration through graded ethanol (Sigma-Aldrich, USA), cleared in xylene (Sigma-Aldrich, USA), and embedded in paraffin wax (Leica Biosystems, Germany) at 58°C. Sections of 5 µm thickness were obtained using a rotary microtome (Leica RM 2135, England), mounted on clean microscopic slides (Marienfeld, Germany), and stained with Hematoxylin and Eosin (H&E) staining kit (Sigma-Aldrich, USA) according to the method of Akpantah et al. (2003).
Statistical analysis
Data were analyzed using GraphPad Prism software (version 10.2.0). All results were expressed as Mean ± Standard Error of Mean (SEM). Statistical differences between groups were evaluated using one-way analysis of variance (ANOVA), followed by Tukey post-hoc. A P-value of less than 0.05 was considered statistically significant.
RESULTS
Effects of 35% carbamide peroxide on body, testicular weights and sperm count in male Wistar rats
There was a slight increase in body weight in the carbamide peroxide groups (100 mg/kg, 250 mg/kg, and 500 mg/kg) compared to the control group (Figure 1). However, these increases were not statistically significant. Additionally, there was a slight increase in testicular weight in the carbamide peroxide groups (100 mg/kg and 500 mg/kg) compared to the control.
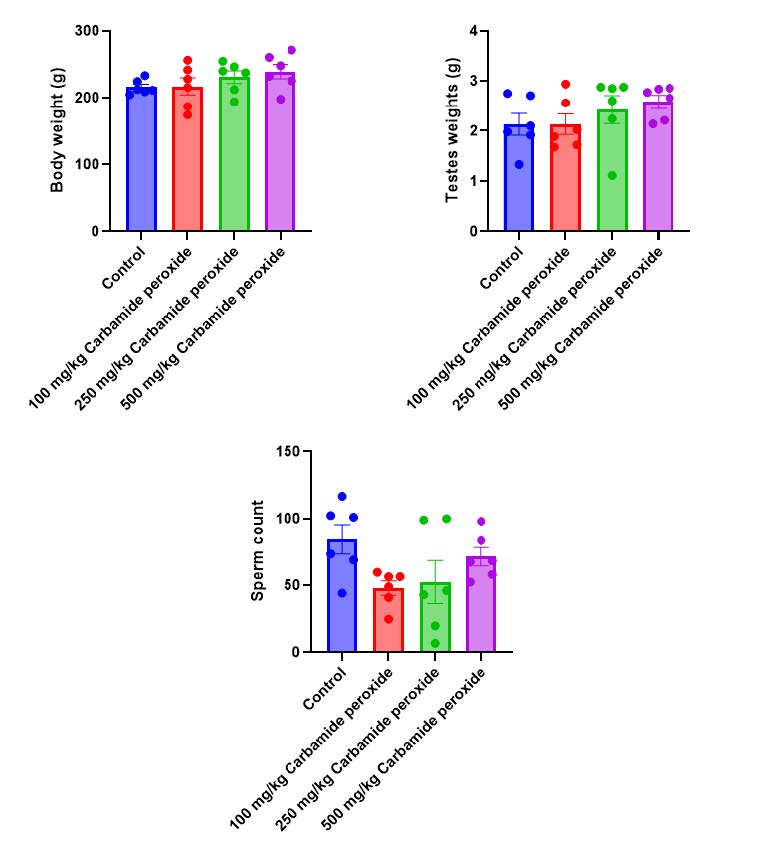
Figure 1. Effects of 35% carbamide peroxide on body, testes weights and sperm count in male Wistar rats. Data were expressed as mean ± SEM (n=6, all groups) using one-way ANOVA followed by Tukey multiple comparison post-hoc test. Values were considered significant at P<0.05*, P<0.01**, P<0.001***, and P<0.0001****.
Effects of 35% carbamide peroxide on sperm motility, morphology and sperm count in male Wistar rats
The results showed a significant decrease (P<0.05) in sperm motility in the carbamide peroxide-treated group (100 mg/kg, 250 mg/kg, 500 mg/kg) compared to the control (Table 1). The carbamide peroxide-treated groups (100 mg/kg and 500 mg/kg) exhibited a 23.5% increase in sperm motility compared to the control group. The percentage increase in non-motile sperm cells in the carbamide peroxide-treated groups (100 mg/kg, 250 mg/kg, and 500 mg/kg) was 16.6%, 44.4%, and 23.1%, respectively. The percentage of normal sperm cells slightly increased by 10% in the 100 mg/kg group and slightly decreased by 30% in the 500 mg/kg group. Abnormal sperm cells increase by 42.86%, 25%, and 12.5% in 100 mg/kg, 250 mg/kg, and 500 mg/kg carbamide peroxide-treated groups compared to the control group. Sperm count decreased in the 100 mg/kg and 250 mg/kg carbamide peroxide-treated groups compared to the control.
Table 1. Effects of oral administration of 35% carbamide peroxide on sperm motility and morphology in male Wistar rats.
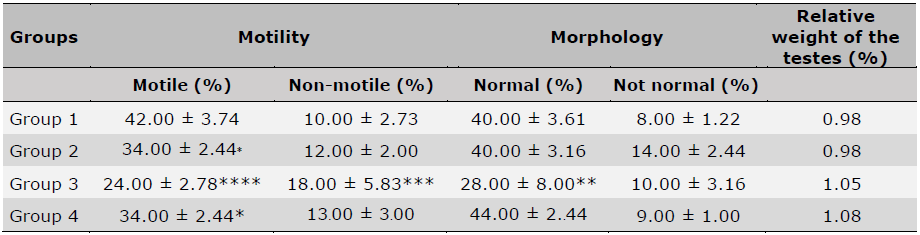
Note: Data were expressed as mean ± SEM (n=6, all groups) using one-way ANOVA followed by Tukey multiple comparison post-hoc test. Values were considered significant at P<0.05*, P<0.01**, P<0.001***, and P<0.0001****. Group 1 (Control) Groups 2 (100 mg/kg carbamide peroxide), Group 3 (250 mg/kg carbamide peroxide), and Group 4 (500 mg/kg carbamide peroxide).
Effects of 35% carbamide peroxide on malondialdehyde, pituitary and male gonadal hormones in male Wistar rats
The results showed a significant increase (P<0.001, P<0.0001) in MDA levels in the carbamide peroxide-treated groups (250 mg/kg and 500 mg/kg) compared to the control (Figure 2). However, there was no significant difference (P>0.05) in MDA levels in the carbamide peroxide-treated group (100 mg/kg) compared to the control. There was a significant decrease (P<0.001, P<0.0001) in FSH levels in the carbamide peroxide-treated group (100 mg/kg and 250 mg/kg) compared to the control group. However, no significant difference (P>0.05) was observed in LH levels in the carbamide peroxide-treated groups (100 mg/kg, 250 mg/kg, and 500 mg/kg) compared to the control group. Testosterone level increased in carbamide peroxide group (500 mg/kg) when compared to the control.
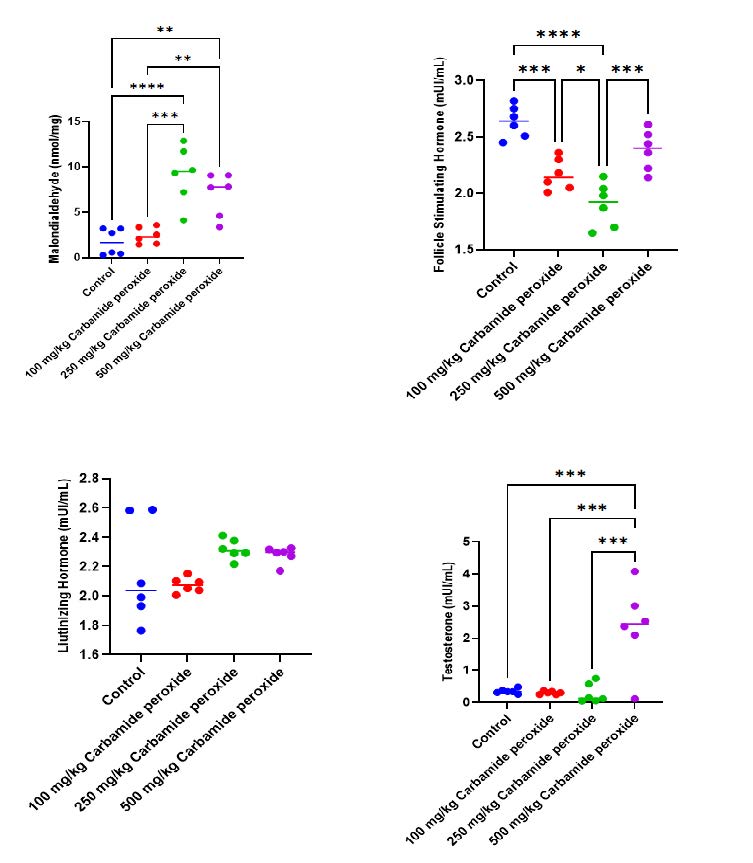
Figure 2. Effects of 35% carbamide peroxide on malondialdehyde, pituitary and male gonadal hormones in male Wistar rats. Data were expressed as mean ± SEM (n=6, all groups) using one-way ANOVA followed by Tukey multiple comparison post-hoc test. Values were considered significant at P<0.05*, P<0.01**, P<0.001***, and P<0.0001****
Effects of 35 % carbamide peroxide on histoarchitecture of the testes and epididymis in male Wistar rats
The photomicrographs demonstrated notable testicular changes in the carbamide peroxide-treated groups (100 mg/kg and 500 mg/kg) compared to the control group. Specifically, there was visible enlargement or hypertrophy of the testes in 100 mg/kg carbamide test group, suggesting that carbamide peroxide may induce abnormal growth in testicular tissue at the dose administered (Figure 3). This hypertrophy could be linked to altered cellular processes, such as increased fluid retention or changes in tissue architecture caused by oxidative stress or inflammation. In addition to testicular enlargement, the photomicrographs of the epididymis revealed increased fat deposition (steatosis) in the carbamide peroxide-treated groups, particularly at doses of 100 mg/kg and 500 mg/kg, compared to the control group (Figure 4). This accumulation of fat in the epididymal tissue could indicate disruptions in lipid metabolism or inflammation within the reproductive system, potentially impairing the normal function of the epididymis, which plays a crucial role in sperm maturation and storage.
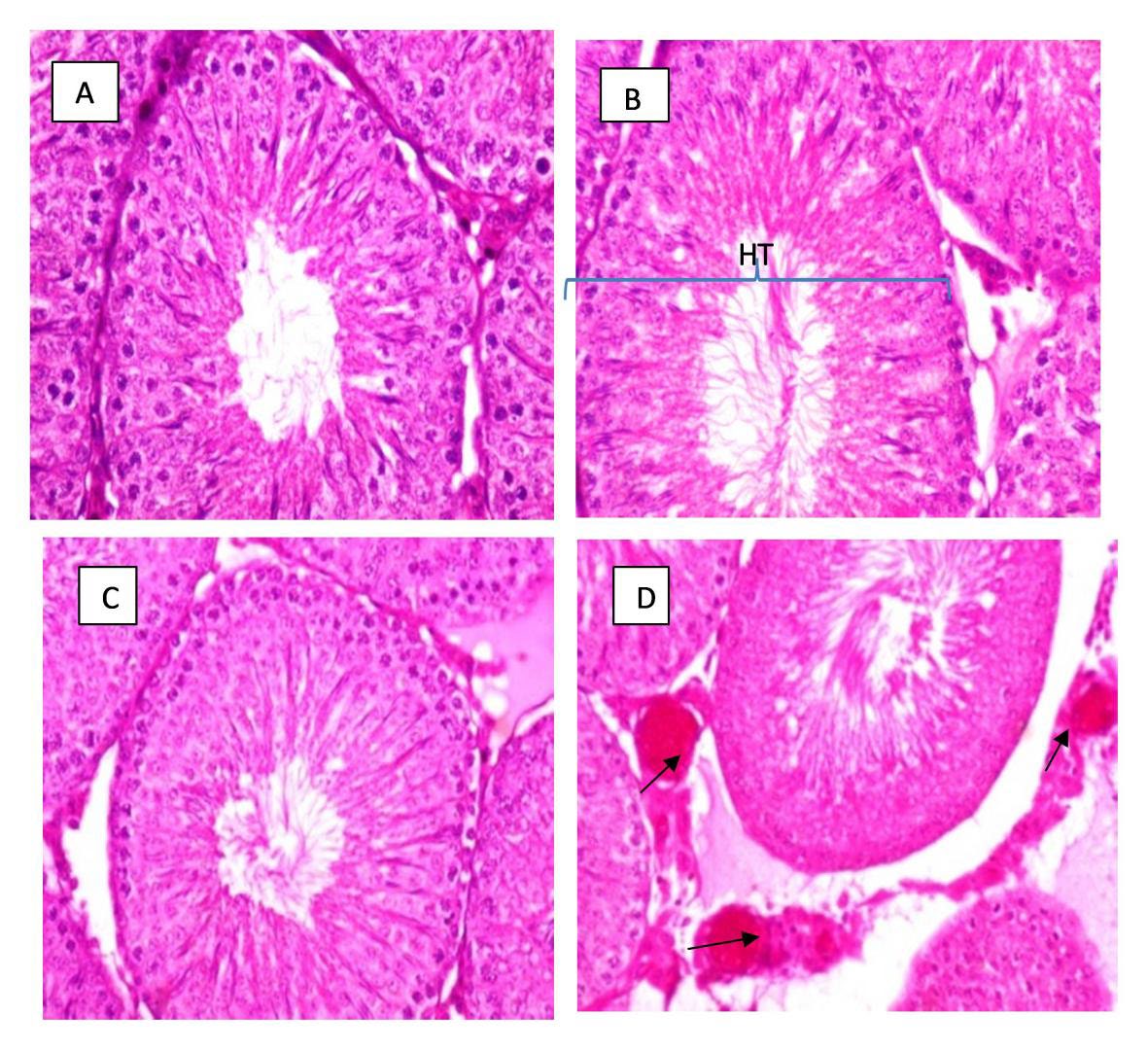
Figure 3. Effects of 35% carbamide peroxide on the testes of male Wistar rats. Figure 3A&C displays the normal architecture of the testes in the control group. Figures 3B show slight hypertrophy (HT) of the testes in the carbamide peroxide-test group at 100 mg/kg. Figure 3D highlights blood vessel damage in the testes of the 500 mg/kg carbamide peroxide-test group (indicated by black arrows). All images were captured at ×400 magnification.
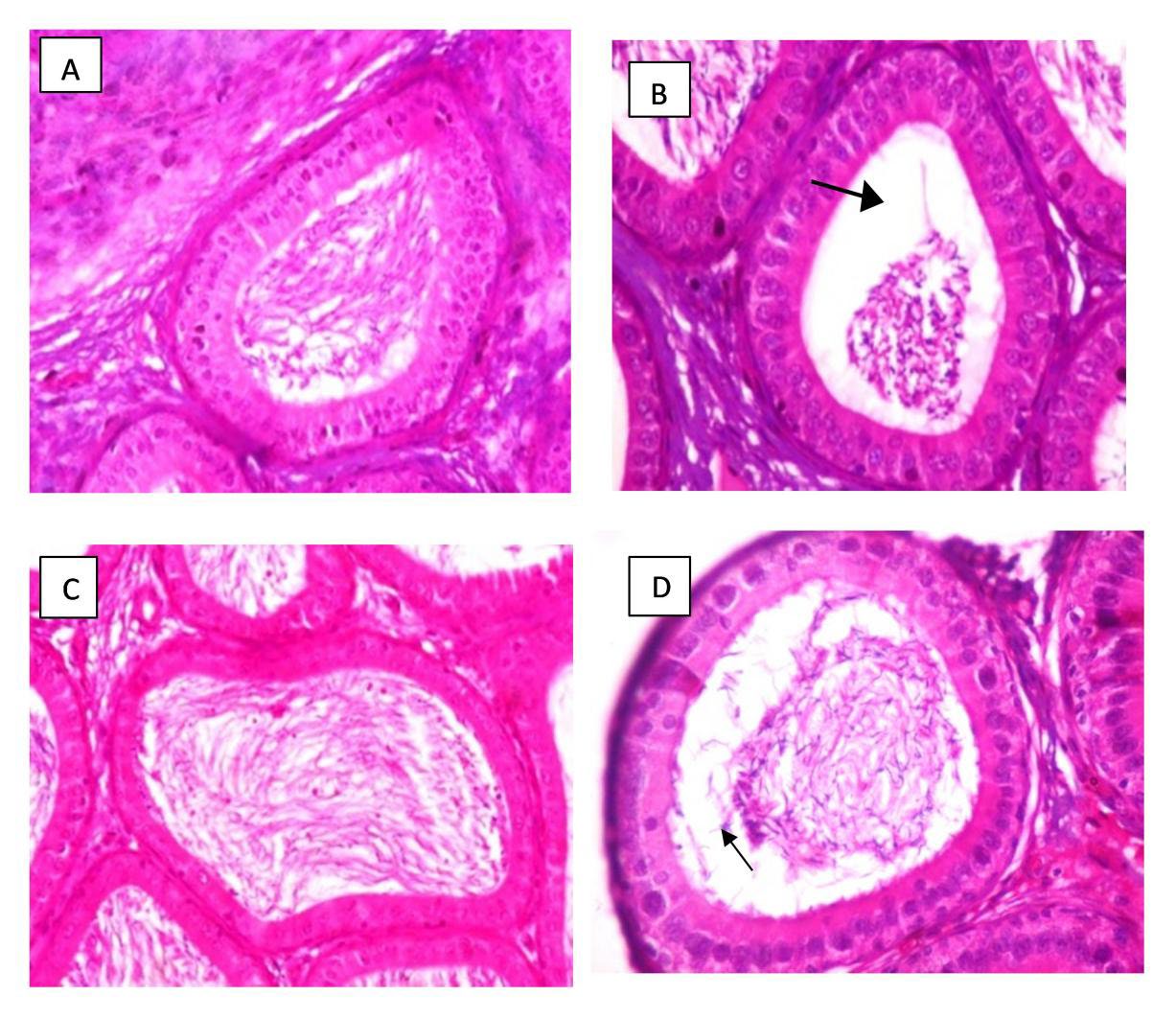
Figure 4. Effects of 35% carbamide peroxide on the epididymis of male Wistar rats. Figure 4A&C displays the normal architecture of the epididymis in the control group. Figures 4B and 4D reveal testicular steatosis in the epididymis of the 100 mg/kg and 500 mg/kg carbamide peroxide-test groups (indicated by black arrows). All images were captured at ×400 magnification.
DISCUSSION
This study investigated the effects of carbamide peroxide, a common active ingredient in tooth-whitening products, on male reproductive function. Tooth whitening has gained widespread popularity as a solution for removing stains caused by food, beverages, and lifestyle habits (Carey, 2014). However, there is increasing concern about the potential adverse effects associated with the use of these products, particularly on systemic health. This study, therefore, aimed to assess the impact of 35% carbamide peroxide on key reproductive parameters, including body and testicular weights, oxidative stress markers (malondialdehyde), reproductive hormones (follicle-stimulating hormone, luteinizing hormone, testosterone, and histopathological changes in the testes and epididymis.
Body weight is influenced by a complex interplay of factors such as age, gender, and genetic predisposition, which are mostly beyond individual control. Previous studies have shown that these intrinsic factors affect metabolic rate, fat distribution, and the likelihood of weight changes over time (Sahin et al., 2023). However, lifestyle and environmental factors, including diet, physical activity, and exposure to chemicals, can significantly modulate body weight. Higaki et al., 2022 highlighted the role of energy balance and metabolic flexibility in body weight regulation, while Hsu et al., 2022 underscored the impact of environmental toxins on body fat and metabolism. These factors can trigger weight gain or loss depending on the exposure and individual susceptibility. In this study, despite the administration of carbamide peroxide, there were no significant changes in overall body weight, which suggests that systemic toxicity was not apparent at the tested doses. This is consistent with findings from other studies, where localized or organ-specific effects can occur without major systemic toxicity, particularly in short-term exposures (Das, 2023). Interestingly, the study observed an increase in testicular weight in the 500 mg/kg carbamide peroxide group. The histological examination revealed steatosis in the epididymis, which is characterized by the abnormal accumulation of fat. This steatosis can result from disruptions in lipid metabolism, and previous research has shown that certain environmental toxins and chemicals can induce lipid deposition in reproductive organs, contributing to organ enlargement (Mima et al., 2018). This localized fat deposition may reflect a tissue-specific response to carbamide peroxide exposure, potentially leading to altered testicular function.
Sperm motility is a critical determinant of male fertility, primarily regulated by calcium ions (Ca²⁺) and cyclic AMP-dependent protein kinase A pathways (Pereira et al., 2017). These signaling pathways are crucial for facilitating two key processes: sperm capacitation, which is the physiological changes sperm undergo to gain the ability to fertilize an oocyte, and the acrosome reaction, where enzymes are released to penetrate the oocyte's protective layers (Rodríguez-Páez et al., 2023). Disruptions in these pathways can lead to impaired sperm motility, significantly affecting fertility potential. In this study, a significant reduction in sperm motility was observed in the group treated with 250 mg/kg of carbamide peroxide. This finding suggests that carbamide peroxide may interfere with the calcium signaling pathways essential for motility and capacitation. Previous studies have established a clear link between exposure to environmental toxins and impaired sperm function. For instance, Seli and Taylor (2023) demonstrated that exposure to endocrine-disrupting chemicals can adversely affect sperm motility by altering hormone levels and signaling pathways critical for spermatogenesis and sperm function. Similarly, Dehdari Ebrahimi et al. (2023) found that exposure to heavy metals resulted in decreased sperm motility and increased DNA fragmentation, underscoring the vulnerability of sperm to environmental contaminants. The observed increase in non-motile sperm cells in this study raises serious concerns regarding the potential reproductive risks associated with the use of carbamide peroxide-containing products (Chittasupho et al., 2023). The implications of reduced motility are profound, as impaired sperm movement can hinder successful fertilization and contribute to male infertility. Collectively, these findings underscore the need for further investigation into the reproductive safety of common chemical agents like carbamide peroxide, especially for individuals seeking cosmetic dental treatments that may inadvertently compromise their reproductive health.
Sperm morphology is a widely used parameter in assessing male fertility, but its reliability is often debated due to the subjective nature of its evaluation (Kumar and Singh, 2015). Variations in the techniques and criteria for analyzing sperm shape contribute to this inconsistency. However, despite its limitations, sperm morphology remains a valuable marker of reproductive health, providing insights into the potential for successful fertilization (Kumar and Singh, 2015). In support of this, previous research has shown that abnormal sperm morphology can impair the sperm's ability to penetrate the egg, reducing the chances of conception (Kondracki et al., 2023; Şişman and Ceylan 2023). Our study further supports these findings by demonstrating that exposure to carbamide peroxide led to a higher percentage of abnormal sperm cells. This increase in abnormal sperm is a key factor that could negatively impact the fertilization process by reducing the sperm’s ability to reach or fertilize the egg. Similar studies have shown that environmental or chemical exposure can induce sperm abnormalities, leading to decreased fertility potential (Zhang et al., 2023). In addition to sperm morphology, sperm count is another essential parameter for assessing male fertility. In our study, carbamide peroxide exposure at 100 mg/kg and 250 mg/kg doses significantly reduced sperm count. This decline aligns with a decrease in follicle-stimulating hormone (FSH) levels, as shown in Figure 2, suggesting that carbamide peroxide may disrupt spermatogenesis by inhibiting FSH production or affecting Sertoli cell function. This conclusion is consistent with the work of Zhao et al., 2023, who also reported that disruptions in FSH levels could impair Sertoli cell function, thereby hindering normal sperm production and reducing fertility potential.
Oxidative stress, defined by an imbalance between free radicals and antioxidant defenses, plays a significant role in male infertility (Pizzino et al., 2017; Osotprasit et al., 2021). In our study, we observed increased levels of malondialdehyde (MDA), a reliable biomarker of lipid peroxidation, in the 100 mg/kg and 250 mg/kg carbamide peroxide groups. This increase in MDA levels suggests oxidative damage to reproductive tissues, as lipid peroxidation can compromise cell membrane integrity and lead to impaired sperm function. Lipid peroxidation results in damage to spermatozoa by affecting their membranes, thus altering motility and viability, which are critical for successful fertilization. Similar studies have shown that oxidative stress, characterized by elevated lipid peroxidation, is a key factor in damaging spermatozoa, leading to fertility issues (Sirisa-ard et al., 2023; Takalani et al., 2023). Further investigation into its impact on other oxidative stress markers are needed to clarify its role in oxidative stress and determine the full extent of its effects on the male reproductive system.
Reproductive hormones, particularly follicle-stimulating hormone (FSH) and luteinizing hormone (LH), are essential for regulating spermatogenesis and testosterone production. These hormones are secreted by the anterior pituitary in response to gonadotropin-releasing hormone (GnRH) from the hypothalamus (Orlowski and Sarao, 2020). FSH specifically plays a critical role in stimulating Sertoli cells, which support the process of sperm development, while LH primarily targets Leydig cells to stimulate testosterone production. In this study, while LH levels remained unchanged, FSH levels were significantly reduced in the carbamide peroxide-treated groups (Figure 2). This reduction in FSH is likely a key factor contributing to the observed impairment in spermatogenesis and reduced sperm count, as FSH is directly involved in promoting the production and maturation of sperm. The importance of FSH in spermatogenesis has been well-established in previous studies, which indicate that disruptions in FSH levels can lead to compromised sperm production and fertility issues (Christin-Maitre and Young, 2022). Despite the reduction in FSH, testosterone levels remained unaffected. This lack of change in testosterone may be explained by the stable LH levels observed, as LH is the primary hormone responsible for stimulating Leydig cells to produce testosterone (Zirkin and Papadopoulos, 2018). This suggests that carbamide peroxide may selectively impact FSH-mediated pathways without significantly affecting LH-driven testosterone production, further emphasizing the specific role of FSH in spermatogenic impairment.
Histological analysis of the testes in the carbamide peroxide-treated groups revealed significant changes, despite the overall testicular architecture remaining intact. One of the notable findings was hyperplasia, particularly evident in the 100 mg/kg carbamide test group, indicating an abnormal increase in size, which could reflect a compensatory response to damage or stress in the tissue. Hyperplasia in testicular tissues can disrupt normal spermatogenesis, as it often leads to alterations in the microenvironment required for sperm development (Teves and Roldan, 2022). Additionally, epididymal steatosis, characterized by fat infiltration into the tissue, was observed in 100 mg/kg and 500 mg/kg carbamide peroxide test groups. Steatosis is uncommon and can impair normal testicular function by interfering with cellular metabolism and hormone production. Studies have shown that lipid accumulation in the epididymis and testes can lead to impaired spermatogenesis and reduced fertility, as the excess fat can disrupt the balance of necessary nutrients and energy required for sperm production (Barrachina et al., 2022). These histopathological changes suggest that carbamide peroxide exposure induces structural alterations in reproductive tissues, which may ultimately compromise fertility. The presence of cellular hyperplasia and fat infiltration underscores the need for further investigation into the specific mechanisms by which carbamide peroxide impacts reproductive health. Similar studies have linked oxidative stress and chemical exposure to histological changes in the testes and epididymis, leading to reduced fertility potential (Ferrandino et al., 2022).
CONCLUSION
This study demonstrated that oral administration of carbamide peroxide at 100 mg/kg, 250 mg/kg, and 500 mg/kg had significant negative effects on male Wistar rats' reproductive health. It reduced sperm count, motility, and normal morphology while lowering follicle-stimulating hormone (FSH) levels. Testicular hyperplasia indicated structural damage, raising concerns about its widespread use in tooth-whitening products and potential fertility risks. However, its relevance to human reproductive health requires clinical validation. This study focused on short-term exposure, leaving long-term effects unclear. The mechanisms affecting spermatogenesis and hormonal regulation remain poorly understood, particularly regarding oxidative stress, hormonal imbalances, and lipid metabolism. Further research is needed to explore these biochemical pathways and assess chronic exposure risks at real-world doses. Human clinical studies are essential to confirm these findings and establish safe usage guidelines for carbamide peroxide-containing products.
AUTHOR CONTRIBUTIONS
Olugbemi T. Olaniyan: Conceptualization, original draft preparation, writing, reviewing, and editing, methodology, data acquisition and analysis; Olorunsola I. Adeyomoye: conceptualization, writing, reviewing and editing, preparation of original draft; Gloria Okotie: conceptualization, writing, reviewing and editing; Femi Adebayo: conceptualization, data validation, reviewing and editing; Ibitoye O Babatunde: conceptualization, writing, methodology, reviewing and editing. All authors have read and approve the submission of this manuscript.
DATA AVAILABILITY
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
CONFLICT OF INTEREST
The authors declare that they have no competing interests.
REFERENCES
Adamkovicova, M., Toman, R., Martiniakova, M., Omelka, R., Babosova, R., Krajcovicova, V., Grosskopf, B., and Massanyi, P. 2016. Sperm motility and morphology changes in rats exposed to cadmium and diazinon. Reproductive Biology and Endocrinology. 14(1): 42.
Akpantah, A.O., Oremosu, A.A., Ajala, M.O., Noronha, C.C., and Okanlawon, A.O. 2003. The effect of crude extract of Garcinia kola seed on the histology and hormonal milieu of male Sprague-Dawley rats' reproductive organs. Nigerian Journal of Health Biomedical Sciences. 2: 40-46.
Albanai, S.R., Gillam, D.G., and Taylor, P.D. 2015. An overview on the effects of 10% and 15% carbamide peroxide and its relationship to dentine sensitivity. The European Journal of Prosthodontics and Restorative Dentistry. 23(2): 50–55.
Asgari, A., Asgari, H.R., Ghorbanlou, M., Dobakhti, F., and Ghorbanian, M.A. 2024. Cerumenolytic effects of carbamide peroxide in patients with ear wax obstruction. Iranian Journal of Otorhinolaryngology. 36(2): 415–420.
Baker, B.I., Torrey, S., Widowski, T.M., Turner, P.V., Knezacek, T.D., Nicholds, J., Crowe, T.G., and Schwean-Lardner, K. 2019. Evaluation of carbon dioxide induction methods for the euthanasia of day-old cull broiler chicks. Poultry Science. 98(5): 2043–2053.
Barrachina, F., Battistone, M. A., Castillo, J., Mallofré, C., Jodar, M., Breton, S., and Oliva, R. 2022. Sperm acquire epididymis-derived proteins through epididymosomes. Human Reproduction. 37(4): 651–668.
Burow, S., Fontaine, R., von Krogh, K., Mayer, I., Nourizadeh-Lillabadi, R., Hollander-Cohen, L., Cohen, Y., Shpilman, M., Levavi-Sivan, B., and Weltzien, F.A. 2019. Establishment of specific enzyme-linked immunosorbent assay (ELISA) for measuring Fsh and Lh levels in medaka (Oryzias latipes), using recombinant gonadotropins. MethodsX. 6: 1473–1479.
Cannarella, R., Condorelli, R.A., Mongioì, L.M., La Vignera, S., and Calogero, A.E. 2020. Molecular biology of spermatogenesis: Novel targets of apparently idiopathic male infertility. International Journal of Molecular Sciences. 21(5): 1728.
Carey, C.M. 2014. Tooth whitening: What we now know. The Journal of Evidence-Based Dental Practice. 14: Suppl, 70–76.
Chittasupho, C., Samee, W., Tadtong, S., Jittachai, W., Managit, C., and Athikomkulchai, S. 2023. Cytotoxicity, apoptosis induction, oxidative stress, and cell cycle arrest of Clerodendrum chinense flower extract nanoparticles in HeLa cells. Natural and Life Sciences Communications. 22(4): e2023057.
Christin-Maitre, S., and Young, J. 2022. Androgens and spermatogenesis. Annales d'Endocrinologie. 83(3): 155–158.
Daniel, J.M., Witty, C.F., and Rodgers, S.P. 2015. Long-term consequences of estrogens administered in midlife on female cognitive aging. Hormones and Behavior. 74: 77–85.
Das, A. 2023. The emerging role of microplastics in systemic toxicity: Involvement of reactive oxygen species (ROS). The Science of the Total Environment. 895: 165076.
Dehdari Ebrahimi, N., Parsa, S., Nozari, F., Shahlaee, M.A., Maktabi, A., Sayadi, M., Sadeghi, A., and Azarpira, N. 2023. Protective effects of melatonin against the toxic effects of environmental pollutants and heavy metals on testicular tissue: A systematic review and meta-analysis of animal studies. Frontiers in Endocrinology. 14: 1119553.
Deshpande, P.S. and Gupta, A.S. 2019. Causes and prevalence of factors causing infertility in a public health facility. Journal of Human Reproductive Sciences. 12(4): 287–293.
Ferrandino, I., Capriello, T., Félix, L. M., Di Meglio, G., Santos, D., and Monteiro, S. M. 2022. Histological alterations and oxidative stress in adult zebrafish muscle after aluminium exposure. Environmental Toxicology and Pharmacology. 94: 103934.
Ferreira Neves Dias, T.D.R.F.Z., Ferreira de Campos, F.U., Turssi, C.P., Botelho do Amaral, F.L., Gomes França, F.M., and Basting, R.T. 2022. Color change after tooth bleaching with ozone and 10% ozonized carbamide peroxide for in-office use. Medical Gas Research. 12(3). 100–106.
GBD 2021 Fertility and Forecasting Collaborators. 2024. Global fertility in 204 countries and territories, 1950-2021, with forecasts to 2100: A comprehensive demographic analysis for the global burden of disease study 2021. Lancet. 403(10440): 2057–2099.
Haidl, G. 2024. Molecular immunology of the male reproductive system. International Journal of Molecular Sciences. 25(4): 1981.
Higaki, S., Inai, R., and Matsuo, T. 2022. Effects of dietary allitol on body fat accumulation in rats. Journal of Nutritional Science and Vitaminology. 68(4): 348–352.
Hsu, C.N., Hou, C.Y., Chang-Chien, G.P., Lin, S., Yang, H.W., and Tain, Y.L. 2022. Sodium thiosulfate improves hypertension in rats with adenine-induced chronic kidney disease. Antioxidants. 11(1): 147.
Huijben, M., Lock, M.T.W.T., de Kemp, V.F., Beck, J.J.H., De Kort, L.M.O., and van Breda, H.M.K. 2023. Clomiphene citrate: A potential alternative for testosterone therapy in hypogonadal males. Endocrinology, Diabetes and Metabolism. 6(3): e416.
Ide, H. 2023. The impact of testosterone in men's health. Endocrine Journal. 70(7): 655–662.
Jin, Y., Paranhos, K.S., Salamone, A., Bongiorno, W., and Brizuela, M. 2024. Internal tooth whitening. In StatPearls. StatPearls Publishing.
Khumsap, S., Tangtrongsup, S., Towiboon, P., and Somgird, C. 2024. GnRH vaccine could suppress serum testosterone in stallion mules. Animals. 14(12): 1800.
Kondracki, S., Górski, K., Iwanina, M., Kordan, W., and Lecewicz, M. 2023. Association between sperm morphology and sperm count of boar semen. Polish Journal of Veterinary Sciences. 26(4): 695–704.
Kumar, N., and Singh, A.K. 2015. Trends of male factor infertility, an important cause of infertility: A review of literature. Journal of Human Reproductive Sciences. 8(4): 191–196.
Li, J., Nan, B., Xu, Z., Chang, H., Xu, S., Ren, M., Zhang, Y., Wu, Y., Chen, Y., Guo, D., and Shen, H. 2023. Arsenic exposure caused male infertility indicated by testis and sperm metabolic dysfunction in SD rats. The Science of the Total Environment. 904: 166838.
Mima, M., Greenwald, D., and Ohlander, S. 2018. Environmental toxins and male fertility. Current Urology Reports. 19(7): 50.
Odetayo, A.F., Akhigbe, R.E., Bassey, G.E., Hamed, M.A., and Olayaki, L.A. 2024. Impact of stress on male fertility: Role of gonadotropin inhibitory hormone. Frontiers in Endocrinology. 14: 1329564.
Orlowski, M. and Sarao, M.S. 2020. Physiology, follicle stimulating hormone. [Updated 2020 Jul 10]. In: statpearls [internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan.
Osotprasit, S., Samrit, T., Chaiwichien, A., Changklungmoa, N., Meemon, K., Niamnont, N., Manohong, P., Noonong, K., Tamtin, M., Sobhon, P., et al. 2021. Toxicity and anti-oxidation capacity of the extracts from Caulerpa lentillifera. Natural and Life Sciences Communications. 20(3): e2021065.
Öztekin, Ü., Caniklioğlu, M., Sarı, S., Selmi, V., Gürel, A., and Işıkay, L. 2019. Evaluation of male infertility prevalence with clinical outcomes in middle anatolian region. Cureus. 11(7): e5122.
Peixoto, A.C., Vaez, S.C., Pereira, N., Santana, C., Soares, K., Romão, A., Ferreira, L.F., Martins-Filho, P., and Faria-E-Silva, A.L. 2018. High-concentration carbamide peroxide can reduce the sensitivity caused by in-office tooth bleaching: A single-blinded randomized controlled trial. Journal of Applied Oral Science: Revista FOB. 26: e20170573.
Pereira, R., Sá, R., Barros, A., and Sousa, M. 2017. Major regulatory mechanisms involved in sperm motility. Asian Journal of Andrology. 19(1): 5–14.
Pizzino, G., Irrera, N., Cucinotta, M., Pallio, G., Mannino, F., Arcoraci, V., Squadrito, F., Altavilla, D., and Bitto, A. 2017. Oxidative stress: Harms and benefits for human health. Oxidative Medicine and Cellular Longevity. 2017: 8416763.
Rodríguez-Páez, L., Aguirre-Alvarado, C., Chamorro-Cevallos, G., Veronica, A.F., Sandra Irel, C.E., Hugo, C.P., García-Pérez, C.A., Jiménez-Gutiérrez, G.E., and Cordero-Martínez, J. 2023. Polyamines modulate mouse sperm motility. Systems Biology in Reproductive Medicine. 69(6): 435–449.
Sahin, Z., Ozcan, M., Ozkaya, A., Canpolat, S., Kutlu, S., and Kelestimur, H. 2023. Percentages of serum, liver and adipose tissue fatty acids and body weight are affected in female rats by long-term Central kisspeptin treatments. Archives of Physiology and Biochemistry. 129(2): 307–315.
Seli, D.A., and Taylor, H.S. 2023. The impact of air pollution and endocrine disruptors on reproduction and assisted reproduction. Current Opinion in Obstetrics and Gynecology. 35(3): 210–215.
Sirisa-ard, P., Pholsonklam, K., Satchachai, A., Tragoolpua, Y., and Kaewkod, T. 2023. Antioxidant, antibacterial activities and cytotoxicity of garlic leaf extract from garlic waste. Natural and Life Sciences Communications. 22(4): e2023059.
Şişman, T. and Ceylan, Z. 2023. The embryotoxicity of alpha-pinene to the early life stages of zebrafish (Danio rerio Hamilton, 1822). Natural and Life Sciences Communications. 22(2): e2023020.
Takalani, N.B., Monageng, E.M., Mohlala, K., Monsees, T.K., Henkel, R., and Opuwari, C.S. 2023. Role of oxidative stress in male infertility. Reproduction and Fertility. 4(3): e230024.
Teves, M.E. and Roldan, E.R.S. 2022. Sperm bauplan and function and underlying processes of sperm formation and selection. Physiological Reviews. 102(1): 7–60.
Vukšić, A., Rašić, D., Žunec, S., Božina, T., Konjevoda, P., Lovrić, J., Bilušić, M., and Bradamante, V. 2023. The effects of simvastatin and fenofibrate on malondialdehyde and reduced glutathione concentrations in the plasma, liver, and brain of normolipidaemic and hyperlipidaemic rats. Arhiv Za Higijenu Rada I Toksikologiju. 74(1): 34–41.
Zhang, Q., Xu, W., Kong, Z., Wu, Y., and Liu, Y. 2023. Cadmium exposure-induced rat testicular dysfunction and its mechanism of chronic stress. Food and Chemical Toxicology. 182: 114181.
Zhao, W., Adjei, M., Zhang, Z., Yuan, Z., Cisang, Z., and Song, T. 2023. The role of GnRH in Tibetan male sheep and goat reproduction. Reproduction in Domestic Animals = Zuchthygiene. 58(9): 1179–1187.
Zirkin, B.R., and Papadopoulos, V. 2018. Leydig cells: Formation, function, and regulation. Biology of Reproduction. 99(1): 101–111.
OPEN access freely available online
Natural and Life Sciences Communications
Chiang Mai University, Thailand. https://cmuj.cmu.ac.th
Olugbemi T. Olaniyan1*, Olorunsola I. Adeyomoye2, Gloria E. Okotie3, Femi Adebayo4, and Ibitoye O Babatunde5
1 Laboratory for Reproductive Biology and Developmental Programming, Department of Physiology, Kwara State University, Malete, Nigeria.
2 Department of Physiology, University of Medical Sciences, Ondo State, Nigeria.
3 Department of Physiology, University of Ilorin, Ilorin, Nigeria.
4 Department of Physiology, Bingham University Karu, Nasarawa State, Nigeria.
5 Department of Anatomy, Federal University of Technology, Akure, Nigeria.
Corresponding author: Adeyomoye Olorunsola Israel, E-mail: oadeyomoye@unimed.edu.ng
Total Article Views
Editor: Sirasit Srinuanpan,
Chiang Mai University, Thailand
Article history:
Received: November 16, 2024;
Revised: March 1, 2025;
Accepted: March 5, 2025;
Online First: March 12, 2025

